Parametric Response Mapping as an Indicator of Bronchiolitis Obliterans Syndrome after Hematopoietic Stem Cell Transplantation
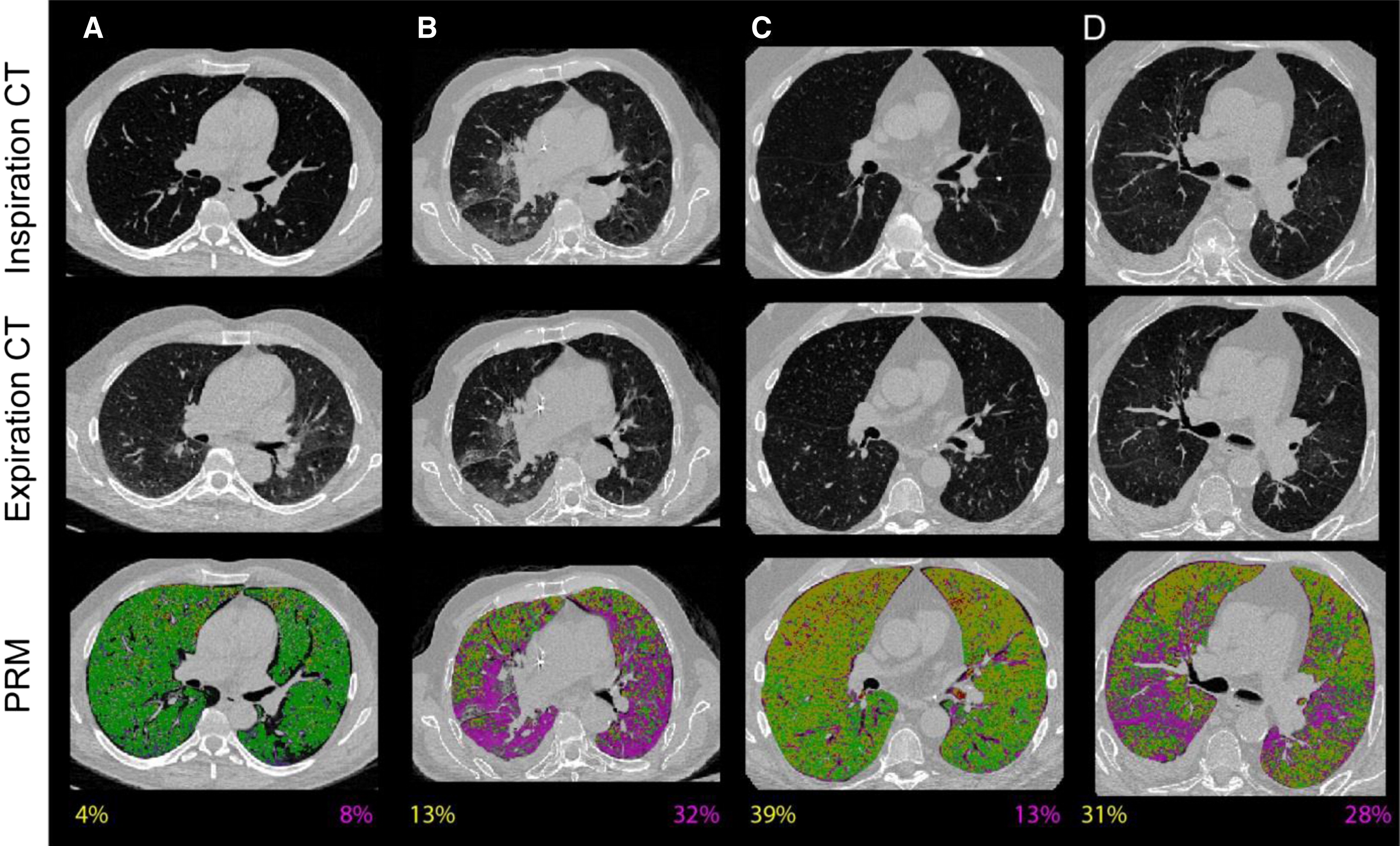
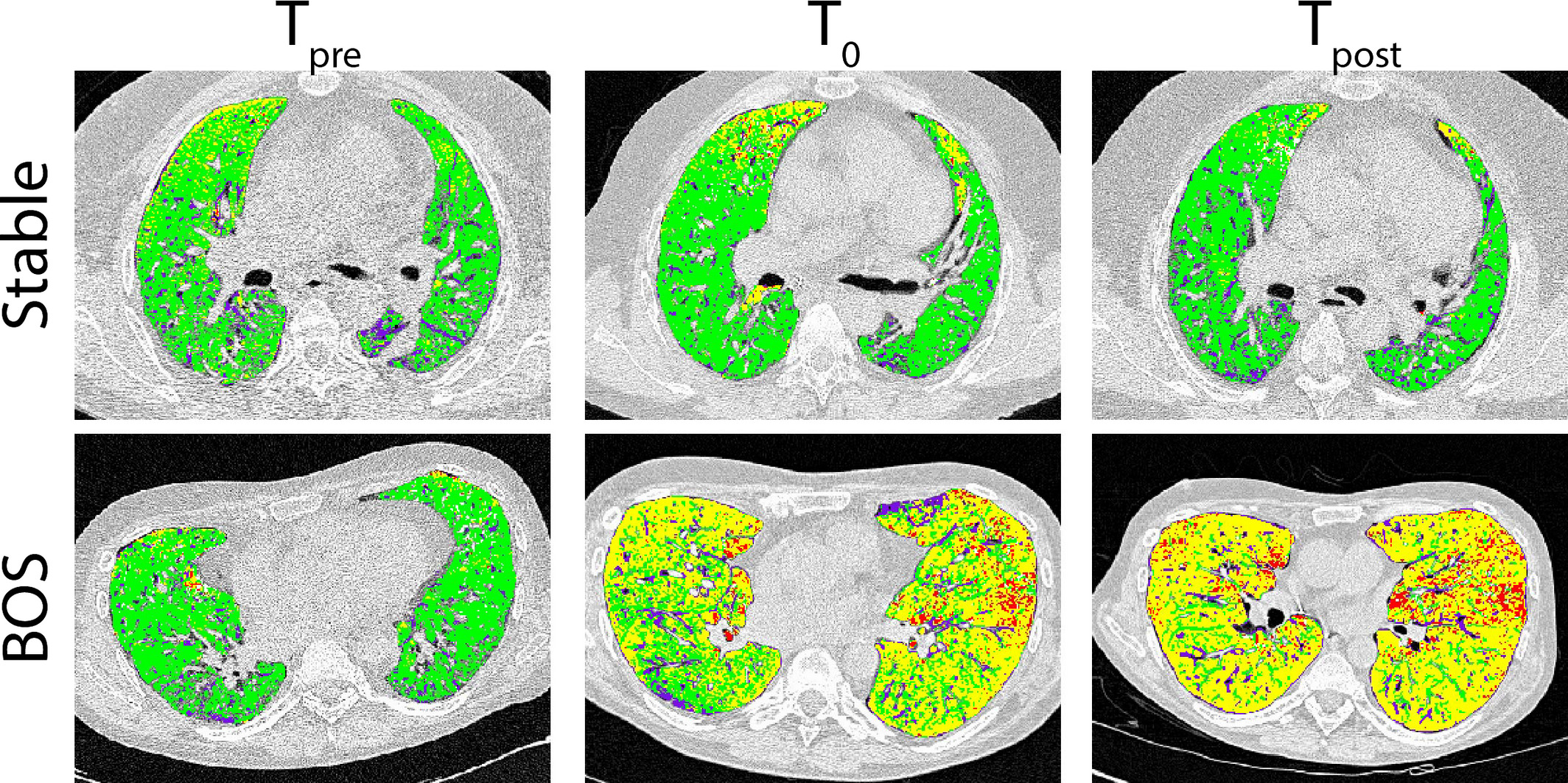
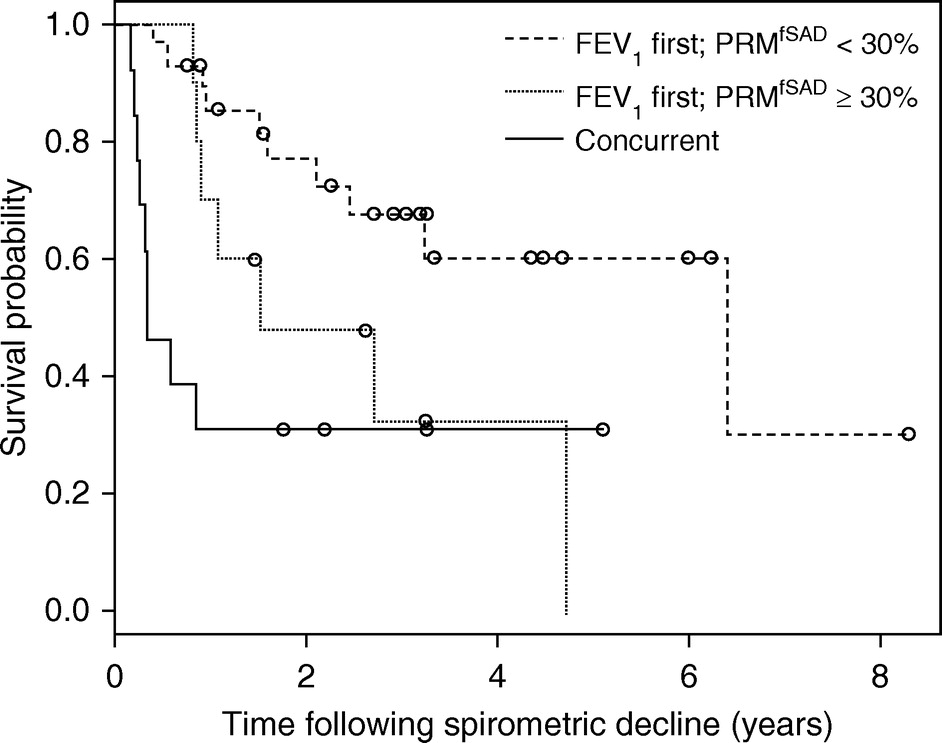
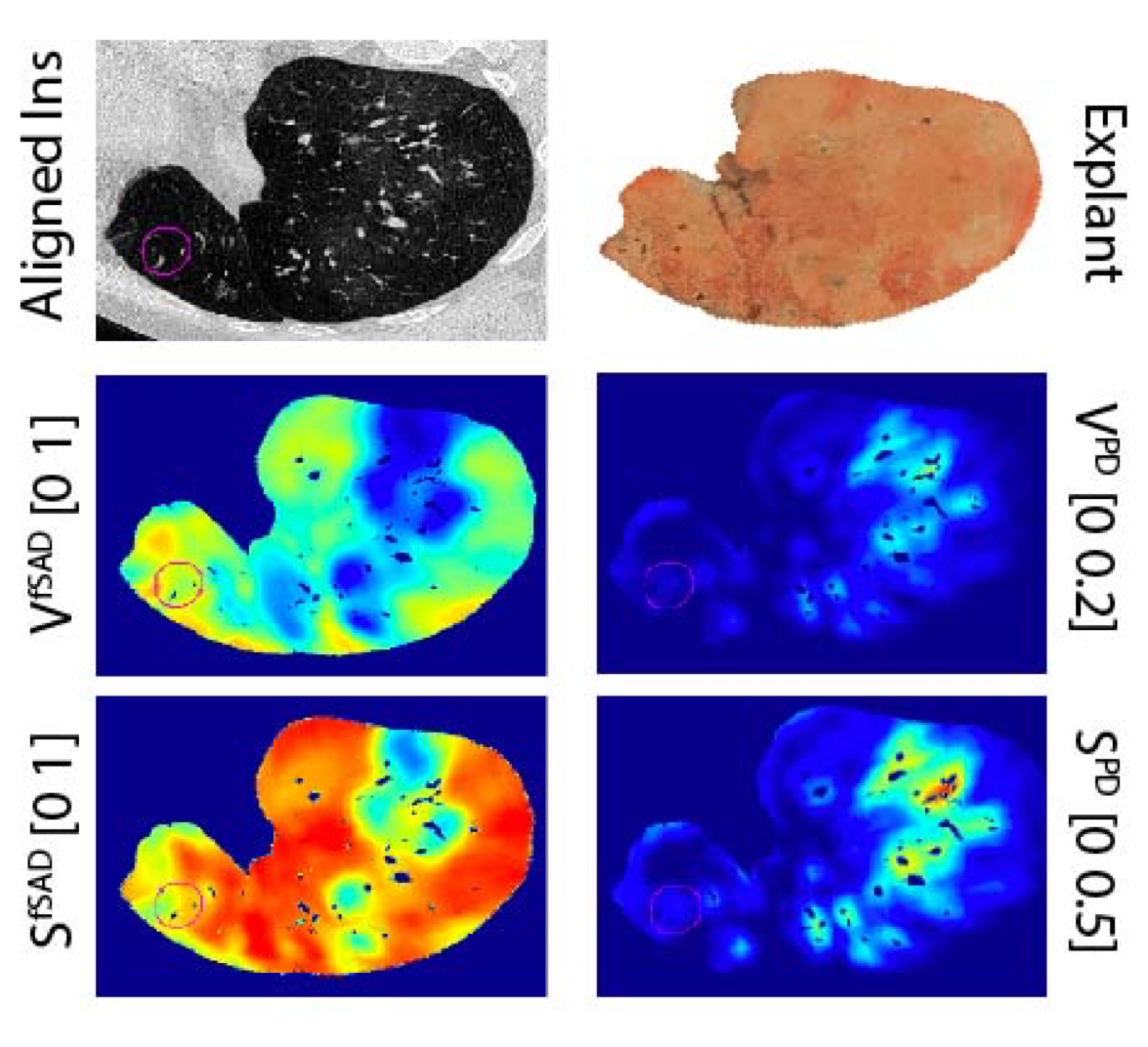
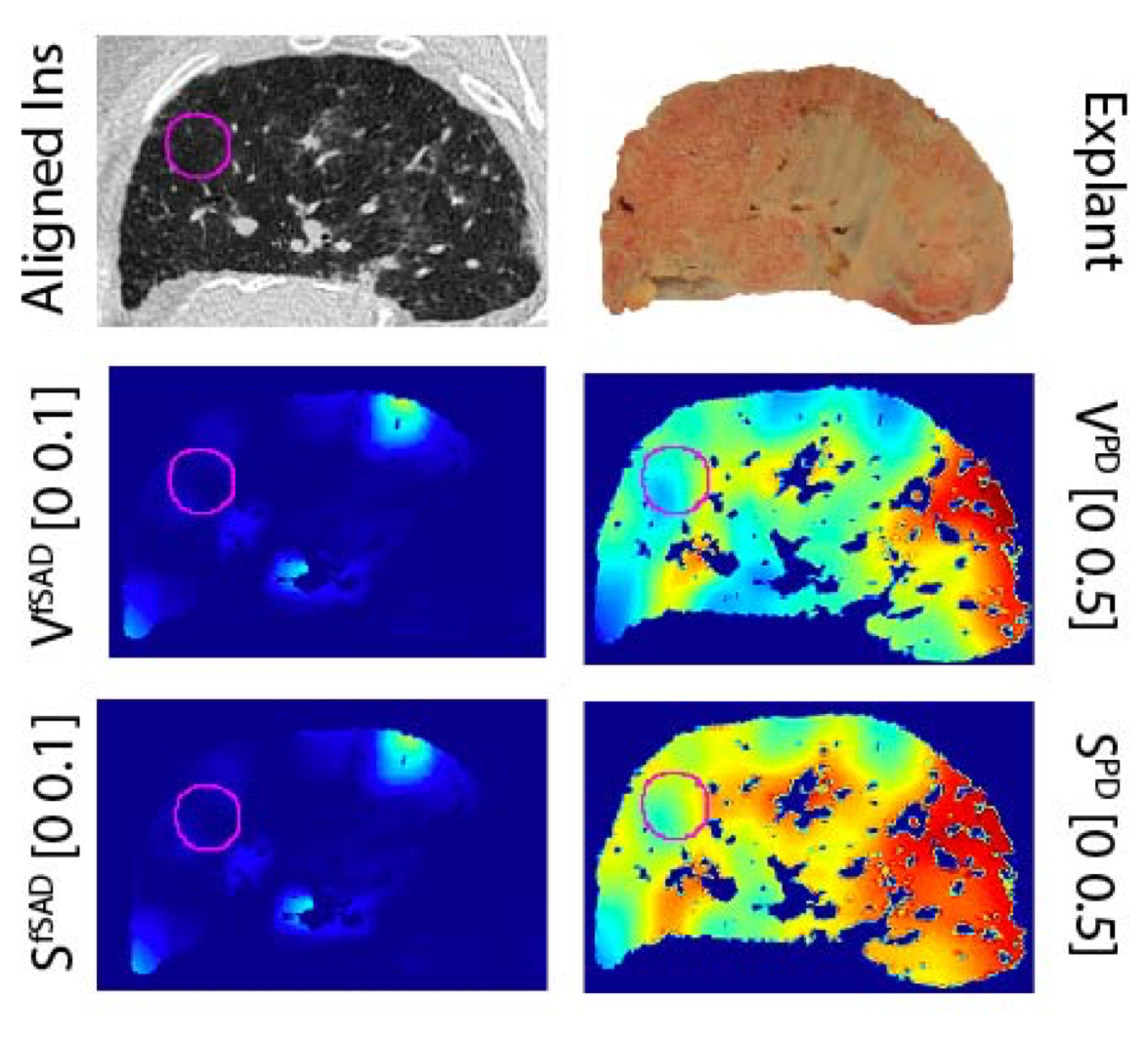
The management of bronchiolitis obliterans syndrome (BOS) after hematopoietic cell transplantation presents many challenges, both diagnostically and therapeutically. We developed a computed tomography (CT) voxel-wise methodology termed parametric response mapping (PRM) that quantifies normal parenchyma, functional small airway disease (PRMfSAD), emphysema, and parenchymal disease as relative lung volumes. We now investigate the use of PRM as an imaging biomarker in the diagnosis of BOS. PRM was applied to CT data from 4 patient cohorts: acute infection (n = 11), BOS at onset (n = 34), BOS plus infection (n = 9), and age-matched, nontransplant control subjects (n = 23). Pulmonary function tests and bronchoalveolar lavage were used for group classification. Mean values for PRMfSAD were significantly greater in patients with BOS (38% ± 2%) when compared with those with infection alone (17% ± 4%, P < .0001) and age-matched control subjects (8.4% ± 1%, P < .0001). Patients with BOS had similar PRMfSAD profiles, whether a concurrent infection was present or not. An optimal cut-point for PRMfSAD of 28% of the total lung volume was identified, with values >28% highly indicative of BOS occurrence. PRM may provide a major advance in our ability to identify the small airway obstruction that characterizes BOS, even in the presence of concurrent infection.
Read Our Paper