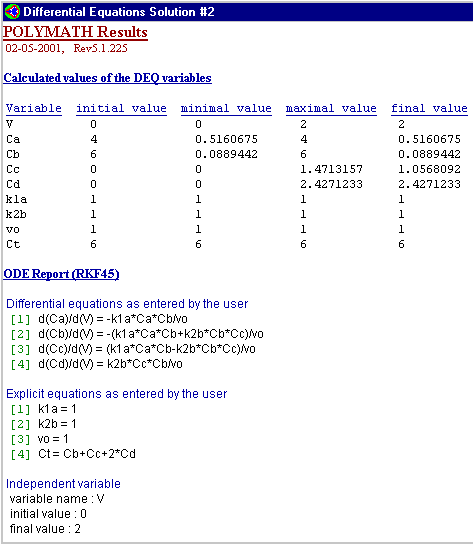
Chapter 8: Multiple Reactions
Concentration-Time Trajectories
1. The following concentration-time trajectories were observed in a batch reactor
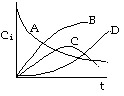
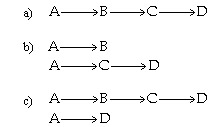
Which of the following reaction pathways best describes the data:
2. Sketch the concentration-time trajectory for the reaction:
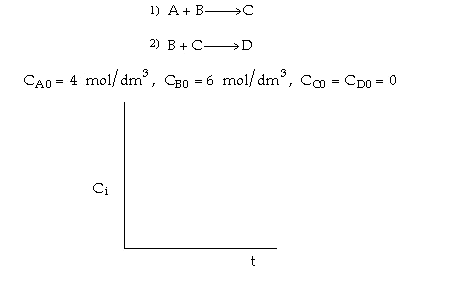
Solution: Part 1
Choice B is the answer. Choices A and C are incorrect because they show species B
eventually consumed, which is clearly not the case.
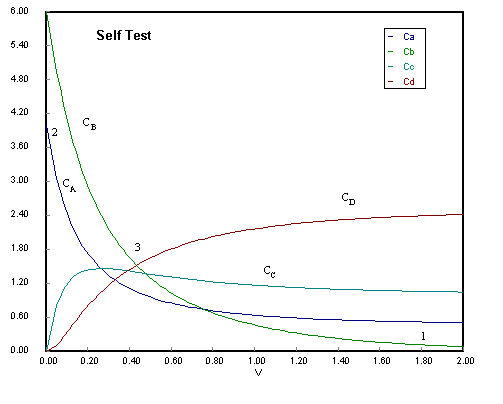
(1) B virtually consumed so no more D can be produced in reaction 2.
(2) Rates of Consumption of A and Ba are virtually the same.
(3) Rate of consumption of B greater than that of A owing to Reaction 2.
Below is the polymath code typed in to produce the graph above.