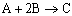 |
(1) | 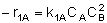 |
NOTE: The specific reaction rate k1A is defined with respect to species A. |
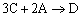 |
(2) | 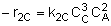 |
NOTE: The specific reaction rate k2C is defined with respect to species C. |
(1) NOTE: The specific reaction rate k1A is
defined with respect to species A.(2) NOTE: The specific reaction rate k2C is
defined with respect to species C.
CASE 1: PFR
CASE 2: CSTR
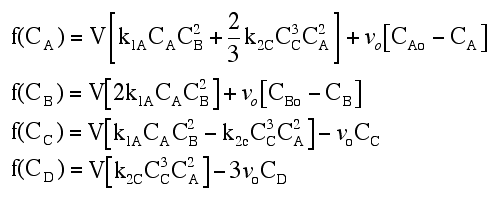
CASE 3: Semibatch
Species A Species B Species C Species D
Evaluate Using Polymath Parameters Initial Conditions