Membrane reactors can be used to achieve conversions greater than the original equilibrium value. These higher conversions are the result of Le Chatelier's Principle; you can remove one of the reaction products and drive the reaction to the right. To accomplish this, a membrane that is permeable to that reaction product, but is impermeable to all other species, is placed around the reacting mixture.
Example: The following reaction is to be carried out isothermally in a membrane reactor with no pressure drop. The membrane is permeable to Product C, but it is impermeable to all other species.
For membrane reactors, we cannot use conversion. We have to work in terms of the molar flow rates FA, FB, FC.


Polymath Program
Mole Balances 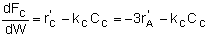



Rate Laws 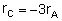
Stoichiometry 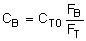



Combine Polymath will combine for you Parameters
 Membrane Type 4 Home Problem (Heterogeneous)
Membrane Type 4 Home Problem (Heterogeneous) Membrane Type 4 Home Problem (Homogeneous)
Membrane Type 4 Home Problem (Homogeneous) Membrane Type 7 Home Problem
Membrane Type 7 Home Problem Membrane Type 8 Home Problem
Membrane Type 8 Home Problem