The reactant that starts in the reactor is always the limiting reactant.
Three Forms of the Mole Balance Applied to Semibatch Reactors:
1. Molar Basis 

2. Concentration Basis 3. Conversion
For constant molar feed: 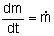


For constant density: Use the algorithm to solve the remainder of the problem.
Example: Elementary Irreversible Reaction
Consider the following irreversible reaction:
The combined mole balance, rate law, and stoichiometry may be written in terms of conversion and/or concentration:
Conversion Concentration Number of Moles Polymath Equations:
Conversion Concentration Moles d(X)/d(t) = -ra*V/Nao d(Ca)/d(t) = ra - (Ca*vo)/V d(Na)/d(t) = ra*V ra = -k*Ca*Cb d(Cb)/d(t) = rb + ((Cbo-Cb)*vo)/V d(Nb)/d(t) = rb*V + Fbo Ca = Nao*(1 - X)/V ra = -k*Ca*Cb ra = -k*Ca*Cb Cb = (Nbi + Fbo*t - Nao*X)/V rb = ra rb = ra V = Vo + vo*t V = Vo + vo*t V = Vo + vo*t Vo = 100 Vo = 100 Vo = 100 vo = 2 vo = 2 vo = 2 Nao = 100 Fbo = 5 Fbo = 5 Fbo = 5 Nao = 100 Ca = Na/V Nbi = 0 Cbo = Fbo/vo Cb = Nb/V k = 0.1 k = 0.01 k = 0.01 Na = Ca*V X = (Nao-Na)/Nao
Polymath Screenshots: Conversion Concentration Polymath Equations Polymath Equations Summary Table Summary Table Conversion vs. Time Conversion vs. Time Concentration vs. Time Concentration vs. Time Volume vs. Time Volume vs. Time