
 |
|
|
|
The reaction rate constant, k, is dependent on temperature according to the Arrhenius equation:
Since the PFR is at a different temperature than the CSTR, we need to relate the two rate constants. The easiest way is by dividing the second rate constant by the first:
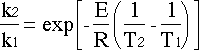
Rearranging gives:
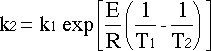
![]() Back to previous page
Back to previous page