
 |
|
Starting with the catalyst differential PFR equation:
Knowing that:
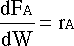
We can rewrite the differential equation:
The differential equation for component B is similar to A except that it is positive because it is a product the rate law:
It must also be taken into account that B is diffusing through the membrane walls. This will change the equation:
The rate will be governed by the equation :
kc prime is the transport coefficient with respect to catalyst weight. We are not given a value for kc prime, but we are given the diffusivity and the boundary layer thickness. Searching through material it can be found that
To find kc prime just divide kc by the catalyst bulk density. Plugging this back into the differential equation for the flow rate of B:
C doesn't diffuse through the membrane so:
![]() Back to previous page
Back to previous page
![]() Part
2 of solution
Part
2 of solution