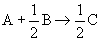

| Remember that the reaction is: |
| For a gas phase system: |
| If the conditions are isothermal (T = T0) and isobaric (P = P0): |
| We must divide by the stoichiometric coefficient of our basis of calculation yielding: |
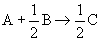 |
| And if the feed is equal molar, then: |
| This leaves us with CA as a function of conversion alone: |
| Similarly for CB: |
 |