
Introduction
Our Delicate Environment:
Finding Ways to
Sustain Civilization
by Joshua Rosen and Michael Schmidt

Introduction
As recorded in photographs and videos, during our first expedition to the moon, we viewed earth from a whole new perspective, as a calm terrestrial planet, made up of an aesthetic blue sphere garnished with swirling white clouds. This image, the image of an uninformed outsider, is extremely deceiving. The truth lies beneath the twirling clouds, the baby-blue tinted sky, and the starry nights. Rather, we must look closer, within our normal localized view, to observe the real, yet disturbing contaminations of earth. Even if the looming global climate change never materializes, the ozone hole remains an equivocal phenomenon, use of toxic air pollutants is reduced, and our consumption of rare and essential natural resources is distributed more efficiently, human activity has already profoundly changed the global environment in ways that we can not perceive from afar. Throughout the world, there are noxious fumes circulating over every major city, radioactive residue stored within our biosphere from nuclear reactions, carcinogenic pesticides harming our agriculture, and deforested habitats destroying the ecology of numerous species. However, these global consequences are in no way recent; most have taken place since the beginning of the earth's history.
Stratospheric Ozone
Multiple environmental issues are associated with a changing atmosphere; of great importance is the depleting ozone layer. The stratosphere, the second highest layer extending about 10-50 kilometers above the earth's surface, is filled with a naturally occurring gas that works as a natural filter-combatting the sun's ultraviolet (UV) radiation called stratospheric ozone.
Please see Figure 1:
The above illustration indicates ozone content on the earth's atmosphere by differentiating between the troposphere, mesosphere, and stratosphere. [Figure taken from http://spso.gsfc.nasa.gov/NASA_FACTS/ozone/fig1.gif]
This ozone originates from interactions between the energetic ultra violet light reaching the stratosphere and molecular oxygen. When this layer of ozone is diminished, more toxic radiation passes through, affecting those on the earth's surface. For humans, an overexposure to UV rays may lead to skin cancer, cataracts and weakened immune systems. From an ecological point of view, an increase in the quantity of these rays results in a deterioration of crops, disturbances within the marine food chain and suppression of photosynthesis. When chlorofluorocarbons (CFCs) such as refrigerants, insulating foams, and solvents diffuse into the atmosphere, they proceed in a process taking as long as two to five years to reach the stratosphere. Once there, the sun's ultraviolet radiation breaks them apart causing the release of chlorine atoms. These chlorine atoms react with ozone, commencing the chemical reactions causing ozone depletion, and hence, destroying the stratospheric ozone layer. One chlorine atom has the potential to break apart up to ten thousand ozone molecules. Pesticides, such as methyl bromide, halons, and methyl chloroform also damage the ozone layer. This process has already created a huge hole above Antarctica. In the lower stratosphere, between 15 and 20 km above the earth's surface, about 95% of the ozone is destroyed. Above 25 km the decreases are smaller and the net result is a thinning of the ozone layer by about 50%. What is essential for us to remember is that, although the ozone hole is currently confined to the Antarctic, its effects are not. For after the hole breaks up in the spring, ozone-poor air drifts north and mixes with the air there, resulting in a transient decrease at middle and high latitudes. Due to its unusual meteorological properties, the Antarctic stratosphere is extremely sensitive to chemical imbalances suggesting that it is extremely unwise to allow CFC emissions to increase by 3% per year, as was occurring during the 1980's. At this rate, increased UV radiation will continually lead to a serious health threat.
Scientists are unsure why this local manifestation of ozone depletion is occurring over Antarctica, but research continues because the ice cap beneath the hole, with an average thickness of 2,500 meters, would raise sea level by 74 meters if it were to melt completely. However, scientists don't even know whether the Antarctic ice sheet is getting thicker or thinner overall. Glaciologist David Vaughan says, "We're completely in the dark. We really have no idea what goes into the ice sheet or what comes out."
Greenhouse Effect
Consider for a moment the earth and it's surrounding atmosphere as a giant greenhouse. Similar to the glass windows of the greenhouse, the earth's atmosphere is somewhat transparent to short-wave and visible solar radiation.
Please see Figure 2:
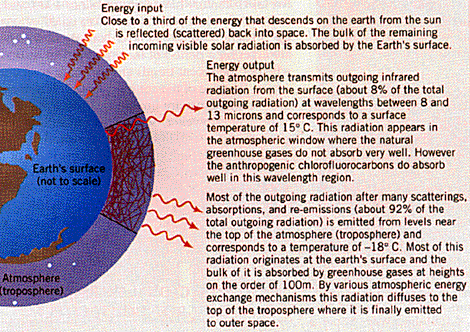
Idealized diagram showing the greenhouse effect. Incoming visible solar radiation is absorbed by the Earth's surface to be re-emitted in the infrared region of the electromagnetic spectrum. Most of the re-emitted infrared radiation is absorbed by the atmosphere maintaining the greenhouse effect. [Caption and figure taken from the text Environmental Science page 432]
Portions of the energy absorbed by the earth radiate back into the atmosphere in the form of long-wave infrared radiation. However, because of the presence of carbon-dioxide and water vapor within the atmosphere, some of the long-wave radiation is absorbed before being re-radiated back to the surface causing the earth's atmosphere to warm. Therefore, slight variations in the quantity of carbon dioxide and water vapor in the air have significant effects on the retention of the earth's atmospheric heat. As a result of the rapid increase in industrial production, we were not concerned with the large amounts of carbon dioxide emitted while burning fossil fuels such as coal, fuel oil, gasoline, and natural gas. When these fuels are burned, carbon is released into the atmosphere, mainly in the form of carbon dioxide, a greenhouse gas that absorbs infrared radiation. "Climatologists have predicted that, as a result of increasing concentrations of greenhouse gases in the atmosphere, the earth's temperature will increase by about three degrees Celsius by the year 2030. Already, global warming has led to an increase in nighttime temperatures in relation to daytime temperatures and increased climatic variability.
Global mean temperature is shown during the years 1880 through 1995. Note that zero is the 1951-1980 average temperature. [Figure taken from http://pubs.acs.org/hotartcl/cenear/951127/img1.html]
For example, heavy rainfall events in the U.S. have gradually increased in intensity while the warming of the eastern equatorial Pacific, El Ni�o, has caused unpredictable intervals of heavy rains and severe droughts.
Please see Figures 4 and 5:
Precipitation abnormalities during El Nino in summer and winter. [Caption and figures taken from http://www.pmel.noaa.gov/toga-tao/el-nino/impacts.html]
Before the advent of a global thermometer, temperatures were measured from a combination of ice cores, ocean sediments, tree rings, fossils, and other geologic data. Providing the most useful information are the ice cores drilled in Greenland and Antarctica that contain "bubbles of air trapped at the time the ice was formed, providing a mechanism to determine atmospheric carbon dioxide concentrations."
Using these devices, one can determine that the earth's temperature will increase by about 3 degrees C by the year 2030 resulting in changes in local climate, loss of arable land, and an increase in sea level with associated coastal flooding. The layers in the ice cores also contain a record of the ocean's temperature in the form of the ratio of isotopically "heavy" water (enriched in 18 O) to "light" water in the ice. When the world's temperature is higher, more heavy water is evaporated preferentially. From the ocean temperature estimate, the air temperature for huge regions can be estimated.
Another telling indicator of the consequences of global warming is the accelerated retreat of alpine glaciers.
Please see Firgure 6 and Figure 7:
Figure 6: A boulder at the western margin of the Quelccaya Ice Cap in the tropical Andes of Peru in 1978. Figure 7: The same boulder in 1995--the extensive retreat of the ice cap in that area has been caused by regional warming. If the warming persists, the ice cap could be in danger of being lost. [Captions and Figures taken from http://pubs.acs.org/hotartcl/cenear/951127/pic1&2.html]
"In the tropics, every glacier that we have any data on is retreating," says Lonnie G. Thompson, professor of geological sciences at Ohio State University. "And where we have time-lapse data, the rate of retreat is accelerating." Countries such as Peru are especially concerned about retreating glaciers. Like many other countries, Peru depends on ice in glaciers to provide irrigation water to the coastal desert and provide water for rivers previously dammed for hydroelectric power. Thus, a loss of glaciers would damage both production and agriculture for the country.
To see a greenhouse effect visualizer visit the following site:
Pollution
Yet another concern to our lifestyle is the continual buildup of harmful pollutants which are attributed mostly to the combustion of coal, oil, and gasoline. Fossil-fuel-fired electric power plants, industrial boilers, and residential furnaces account for more than 80% of the sulfur dioxide, 50% of the nitrogen oxides, and 30 to 40% of the particulate matter emitted to the atmosphere in the U.S. Everyday behavior such as driving a car or truck, results in more than 80% of the carbon monoxide and 40% of the nitrogen oxides and hydrocarbons circulating around us. The tall smokestacks, supposedly projecting polluting chemicals far away from us, only help to spread these toxic materials over a greater distance in order to reduce their concentration at that site.
Carbon dioxide concentrations over the past 1,000 years determined from ice core records (shown as symbols) appear to have fluctuated little until 1850. Since 1958, air measurements (shown as purple line) taken at Mauna Loa, Hawaii, have supplemented the ice core data. The smooth black curve is based on a 100-year running mean. The inset of the period from 1850 onward shows carbon dioxide emissions in gigatons (billions of metric tons) per year attributed to burning fossil fuels (shown as a blue line). [Caption and Figure taken from http://pubs.acs.org/hotartcl/cenear/951127/img2.html]
Doing this ensures transportation of these pollutants, eventually causing adverse effects far away. For example, sulfur dioxide and nitrogen oxide from the Midwest and eastern shore of the U.S. produce acid rain in New York State, New England, and eastern Canada inducing change in the pH level, or relative acidity, of numerous freshwater lakes in that region. With a change in relative acidity, the ecology of marine life is altered, thus killing entire fish populations.
Natural Hazards
The general public, often ignorant when dealing with the effects of natural pollutants on our health, believes that only the factories or large diesel engines cause contamination to our atmosphere. However, it is interesting to learn that many natural processes including dust storms, floods, and volcanic processes, present various harmful effects to humans, animals, agriculture, water, and air as well. For example, on August 21, 1986, when a subaqueous volcano erupted at Lake Nios in Camaroon, Africa, a damaging gas, probably consisting of mostly carbon dioxide, was released and eventually descended upon the villagers. This gas, heavier than air, caused approximately 2000 human deaths by asphyxiation and multiple animal fatalities. Another example, perhaps more relevant to us in the U.S. is radon gas, a naturally occurring colorless, odorless, and tasteless radioactive gas known to cause lung cancer. Radon is produced during the radioactive decay chain from radiogenic uranium to stable lead. The EPA reports that there are approximately 140,000 total lung cancer deaths in the United States each year and attributes about 15% of those deaths to radon gas.
Please see Figure 9:

How radon gas may enter homes. (1) Radon in groundwater enters well and goes to house, where it is used for water supply, dish washing, showers, and other purposes. (2) Radon gas in rocks and soil migrates into basement through cracks in foundation and pores in construction. (3) Radon gas is emitted from construction materials used in building the house. [Caption and figure taken from Environmental Science page 486]
Chemists believe that the health risk associated with radon gas is primarily due to its daughter products, primarily polonium 218, a particle that adheres to dust. Supposedly, when that dust is inhaled into the lungs, cell-damaging alpha radiation occurs with the decaying of Polonium 218. With this reoccurring decay, human DNA strands at the cell level are eventually broken, leading to lung cancer.
Please see Figure 10:

(a) Deposition of Polonium 218 in the lungs. (b) Alpha radiation (particle) breaking one or both DNA strands at the cell level. [Caption and figure taken from Environmental Science page 485]
Nuclear Energy
With 20th century trends leading closer and closer to the use and abuse of nuclear energy,we must concern ourselves with yet another potential problem, the effects of nuclear side-products. Affecting the environment in two major ways, radioisotopes emit radiation harmfully affecting water, agriculture, and atmosphere, and disrupt the normal cycle of the ecological food chain. When a nuclear weapon explodes, many types of intense radiation generating large amounts of radioactive isotopes are released into the environment. A major problem resulting from nuclear testing in the United States, the former Soviet Union, China, France, and Great Britain, during the 1950's and 1960's, was that of atomic fallout, the deposit of these radioactive materials around the world.
Please see Figure 11:

How radioactive substances reach people. [Caption and figure taken from Environmental Science page 361]
During this time, atomic explosions sent radioactive isotopes into the stratosphere leading to an unfortunate example of the disruption of our ecological cycle. One major isotope, that of Cesium 137, was released in the Arctic, but due to wind forces it became dispersed throughout North America. This dispersion first affected reindeer moss, a food of the caribou, but ultimately affected northern Eskimos whose primary source of protein is caribou. Eskimos in turn began to build up potentially harmful high percentages of cesium within their bodies. The major problem presented here is that: "After entering specific ecosystems through the vegetation, [the cesium] underwent biomagnification, or ecological food-chain concentration. That is, at each level of the ecological food chain, the concentration of toxic material relative to concentrations of other materials in the bodies of organisms increased."
Please see Figure 12:
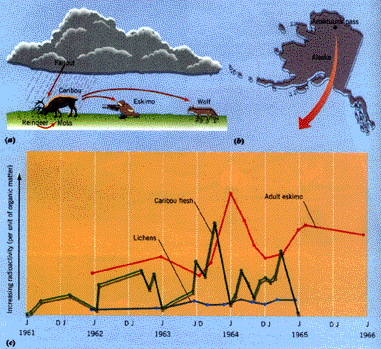
Cesium 197, released into the atmosphere by atomic bomb tests, was part of the fallout deposited onto the soil and plants. (a) The cesium fell on lichens, which were eaten by the caribou. The caribou were in turn eaten by Eskimo. (b) Measurements of cesium were taken in the lichens, caribou, and Eskimo in the Anaktuvuk Pass of Alaska. (c) The cesium was concentrated by the food chain. Peaks in concentrations occured first in the lichens, then in the caribou, and last in the Eskimo. [Caption and figure taken from Environmental Science page 362]
Currently, the future of nuclear energy as a power source for electricity is being vigorously debated. Advocates for nuclear energy argue that nuclear power is beneficial for the environment because it does not potentially induce global warming, nor does it cause air pollution or emit precursors of acid rain. Also, with the availability of breeder reactors, the amount of fuel available will be greatly increased. However, even a greater majority are concerned about possible accidents and waste disposal problems.
Although the chances of a disastrous nuclear accident is very low, the probability of an accident occurring increases with every reactor put into operation. Two of the greatest nuclear disasters occurred within the past twenty years. On March 28, 1979 at the Three Mile Island nuclear power plant near Harrisburg, Pennsylvania, malfunctions of the power plant resulted in the release of radioisotopes into the environment as well as intense radiation release within one of the nuclear facilities. By the third day following the release, radiation levels near the sight at ground level were estimated at 12 mSv/hour. By comparison, the average American receives about 1.5 mSv/year from natural radiation. One of the major impacts was fear and since the long-term chronic effects of exposure to low levels of radiation are not well understood. Currently, scientists are still unable to estimate all of the effects from Three Mile Island.
Please see Figure 13:

The Three Mile Island Nuclear Power Plant--Unit One near Harrisburg, Pennsylvania. [Figure taken from http://www.nrc.gov/AEOD/pib/reactors/289/289toc.html]
Another example of the world's lack of preparedness to deal with nuclear accidents occurred on the morning of April 28, 1986 when workers at a nuclear power plant in Sweeden, frantically searching for the source of raised levels of radiation, determined that there installation was not leaking, but rather, the radioactivity was coming from the Soviet Union by way of prevailing winds. After being confronted, the Soviets announced that there had been an accident at their nuclear plant in Chernobyl. In the days following the accident, there were 237 confirmed situations of acute radiation sickness, 31 people died of radiation sickness, and nearly 3 billion people in the Northern Hemisphere received varying amounts of radiation from Chernobyl. Estimates of the cost of this accident vary widely; low end estimates are $1.6 billion were high end estimates are $200 billion.
Since the Chernobyl incident, many countries in Europe have been reevaluating the use of nuclear power plants, and in most instances, the quantity of new plants being built has reduced. Since 1978, there has been no new orders for nuclear power plants in the United States. However, research and development into the smaller, safer plants is going forward. Nuclear plants produce about one-fifth of electricity in the United States today, and production is expected to increase slightly in the next few years.
Evaluating Our Environment
In his environmental best-seller, Barry Commoner poses the question: "What can be done to end the war between nature and man, between the ecosphere and the technosphere?" As we continually contaminate or prolong initiating precautions protecting us from these detrimental threats, this question will continue to rouse more and more debate during upcoming years. Some are willing to sacrifice future human growth in the way of technological development for the reduction in environmental attack. Others proclaim that a human has the right to extract all of the earth's resources to their fullest, making himself most comfortable at the given time, but deteriorating the earth for future generations. More recently, as large industrial corporations begin to gain political power through lobbying Congress, the outcome of this argument becomes more and more dependent on the money involved. That is, the larger, more affluent corporations can afford to heavily lobby the Congresspeople that vote on environmental laws, whereas the common individual U.S. citizen cannot make nearly as substantial an impact. However, there are a large number of environmental lobby groups including the Sierra Club, the National Wildlife Federation, and the National Reserve Defense Council. Unfortunately, "in the United States, democracy stops at the door to the corporate boardroom." However, this "tightly bound door" can be opened if our government opts to use its massive purchasing power, potentially resulting in fairer economically and environmentally efficient laws.
Taking Action on Pollution
When researching and evaluating the effects of these pollutants and threats to our earth, one must not only concentrate on the negative aspects, but must propose solutions and most importantly realize what can and has been done to sustain our civilization. Perhaps the most influential organization in regulating and controlling pollution has been the EPA (Environmental Protection Agency). In the U.S., the Clean Air Act of 1967 as amended in 1970, 1977, and 1990, specifying that air-quality standards must be set for hazardous substances, is the legal basis for air-pollution control throughout the United States. Emission standards are specified to limit the discharge of pollutants into the air so that air-quality standards can be achieved. The specially amended portion in 1990 specified ozone, carbon monoxide, particulate matter, acid rain, and air toxins as major air pollution problems.
Please see Figure 14 for a listing of the major air pollutants:
Sources of major air pollutants include individual actions, such as driving a car, and industrial activities, such as manufacturing products or generating electricity. [Caption and figure taken from "Major Air Pollutants," Microsoft Encarta ® 96 Encyclopedia. © 1993-1995 Microsoft Corporation.]
Slowing Down Ozone Depletion
The failure to prevent the continual deterioration of the environment has gradually become more and more evident on the global scale. Ozone depletion has continued to worsen and few until the Reagan administration had attempted to fight it. Initially, Reagan's administration attempted to promote the use of dark glasses and suntan lotion, but since then, some positive steps have been taken: An international agreement (The Montreal Protocol) to reduce CFC production by 50% has been reached. Originally signed in 1987 and substantially amended in 1990 and 1992, the protocol stipulates that CFCs, halons, carbon tetrachloride, and methyl chloroform should be virtually extinct by the year 2000. The signing of this landmark protocol established many local reduction laws such as those in Irvine, California, restricting CFC emissions. In August 1989, several automobile companies promised to reduce the emissions of CFCs from automobile air conditioners. More recently, the Dupont chemical company announced the development of a less damaging alternative for CFCs. Another possible solution to lower emissions of CFCs into the atmosphere is to develop ways to collect and reuse CFCs, but with the impeding prohibition on CFCs, we must search for substitutes. Two substitutes for CFCs being experimented with today are hydrofluorcarbons (HFCs) and hydrochlorofluorocarbons (HCFCs). Although controversial and still experimental these substitutes do have some advantages: HFCs do not contain chlorine, but do contain fluorine. However, fluorine has been proven to be approximately 1000 times less efficient in causing ozone depletion than chlorine. In place of a chlorine atom, HCFCs contain a hydrogen atom, allowing them to break down in the lower atmosphere, before reaching the upper stratosphere, preventing the injection of chlorine into the stratosphere. HCFCs are still considered controversial because at large concentrations, they cannot be broken down fast enough and are a threat to depleting the ozone layer. Eventually, if the ozone depleting chemicals are banned, then sometime in the next century ozone depletion should reverse. However, in the near future, the best approach that one can take is to adapt to an increased exposure of ultraviolet radiation.
Global Warming and Deforestation
Like all other environmental issues, debaters struggle over how to approach global warming. Given the information available to us from scientific research, we are faced with two major options: mitigation (reducing the severity of the problem through reduction of emissions of greenhouse gasses) or remaining indifferent and adapting to future global change. Unlike ozone depletion, scientists still do not have sufficient information pertaining to the potential effects of global warming. "More research is needed, but is unlikely to be funded by the U.S. In fiscal 1995, federal agencies spent a total of $1.8 billion on global change research. A somewhat larger amount was requested for 1996, but Congress had proposed substantial cuts in the programs of some agencies." The current estimation for mitigation would have to be in the range of a 20 to 50% reduction in carbon dioxide. Because close to 70% of the anthropogenic carbon dioxide emissions result from the burning of fossil fuels, energy planning relying more heavily on energy conservation and efficiency must include the use of alternative energy sources. These include solar power, wind power, geothermal power, or in some cases nuclear power to reduce emissions. Other options include increased taxes for using fossil fuels, thus providing an incentive to produce both economic and environmental efficiency. Some have even suggested requiring more stringent fuel-economy standards for automobiles and requiring higher standards of energy efficiency for appliances and buildings. By the year 2000, this will be the trend of California's standards.
In some areas, due to increased commercial development, trees are increasingly being chopped down leading to serious problems of deforestation. Chopping down tress results in additional carbon dioxide accumulation because of a reduced occurrence of photosynthesis (a process which uses carbon dioxide to produce oxygen). Burning down forests to convert land to agricultural or commercial purposes roughly account for 20% of the anthropogenic carbon loading into the atmosphere. Often to counter deforestation, governments have set up special areas to plant additional trees (reforestation). Thus, scientists along with economists recommend that the best strategy until more information is known concerning global warming "is somewhat between full mitigation and learning to adapt to change."
Harnessing the Ocean's Energy
While we as humans have mainly used the land as our means of resources for energy, we have an abundant amount of promise in adapting the ocean for future use. The ocean covers two-thirds of our earth, yet we know and use so little of it. While traditional fossil fuels will most likely run out in the next century, the ocean is continuously being replenished. From the ocean a various amount of sources are available to use as new means of energy.
Tidal energy can be dated back hundreds of years ago. It was used by Europeans, Russians, and even along the Atlantic coast of North America. By using the vertical rise and fall of the tides, and the ebb and flood of the tidal currents, and applying a dam across a tidal basin, the rise and fall of the water will drive a generator. This energy can create a worldwide potential of 3 million MWe. A drawback to using this means is the length of the dam needed to close off the basin, has a large cost to build. The environmental impact is very minimal, some of the sandbanks are relocated, and some of the existing fish species may disappear, but they are replaced by new ones.
The harnessing of wave energy has also been used for quite some time. This type of energy can be captured in several ways, by using turbines activated by waves, or by air columns, while relative-motion devices provide the power. An example of this comes from the Lockheed Corporation in 1979. Using a whirlpool which acts as a flywheel, water from the waves spills into the center of a turbine. In an air column, the water rises and falls, which drives a generator. These dam-atolls are, once again, very expensive to build. Costs are estimated at $8,000-$18,000 per kW.
Although most of the ocean's energy is considered as solar energy, the most direct harnessing of ocean solar power is through a plant which uses OTEC-Ocean Thermal Energy Conversion. The basic principle is that in some areas of the world, deep cold ocean water ascends to relatively shallow depths, while in these same areas the surface waters are quite warm from collected solar heat. OTEC uses this temperature difference to power a turbine and generate electricity. This system has the potential of meeting a major portion of America's energy needs. The problems that face OTEC are biofouling and corrosion of its pipes.
Finally one of the most interesting approaches comes from biomass energy. Marine plants, which are the main "receivers" of solar energy in the ocean, can be used as a source of fuel, similar to methane or liquid hydrocarbons. Since marine plants are more productive than land plants, biomass can be a highly effective energy source. It is estimated that 14,245 km^2 (55,000 mi^2) of sea surface could provide all of the gas needed by the United States. Giant kelp could be grown on offshore submerged floats. The basic principle is to grow kelp, using nutrients brought up from the deep, harvest them, and have them anaerobically digested to methane. Because solar energy is always available, vegetal biomass can be changed to energy without ever running out of a supply.
Closing Remarks and Conclusion
Although our modern civilization has had many negative effects on the environment, we must also recognize the many benefits that come with advanced technologies. Whether we like it or not, our society is run through technology. Through these once unperceivable advances, technologically able humans have had an advantage over those without the means. But technology used is powered by some resource, and that resource must come from our environment. For example, humans will continue exploiting the fossil fuels until they run out. We must learn to change our ways of exploiting the earth, to maintain a proper balance with ourselves and our earth. After all, we are in the midst of war between two worlds, the ecosphere and the technosphere. In many ways nature has been devastated causing human suffering, not only because of the depleted ozone layer, global warming, and toxic fumes, but because of our environmental ignorance, diminishing the opportunities for economic growth, especially within our well-developed country. What is needed is a "peace talk that takes into account both nature's need for self sustenance and the human need not only to maintain our present level of material welfare but to increase and disseminate it." Most importantly we must acknowledge that only we can initiate change. Although virtually impossible to control the attack on the environment, it is our duty to ensure a proper balance between economic and environmental efficiency by preventing future environmental degradation. This necessitates an enormous redesigning of the major industrial, agricultural, energy, and transportation strategies. "What is done in the United States and other industrial nations must be compatible with the global task of closing the economic gap between the rich north and the poor south." In other words, we must reduce the generation of toxins while at the same time continue the production of necessary living essentials. Not an easy process, this will take time, great effort and careful research, but with all of humankind's current day accomplishments, no task has yet proven to be impossible.
Glossary
acid rain- rain made artificially acid by pollutants, particularly oxides of sulfur and nitrogen
air toxins- total suspended matter found in the atmosphere as solid pieces or liquid droplets; airborne particulates include windblown dust, emissions from industrial processes, smoke from the burning of wood and coal, and the exhaust of motor vehicles
anaerobic- characterized by the absence of free oxygen
anthropogenic- man-made, usually used in the context of emissions that are produced as the result of human activities
atomic fallout- the deposit of radioactive materials around the world
biofouling- fouling caused by growth of marine organisms (organic slimes, algae, encrusting organisms)
biomagnification- the tendency for some substances to concentrate with each tropic level; organisms preferentially store certain chemicals and excrete others; when this occurs consistently among organisms, the stored chemicals increase as a percentage of the body weight as the material is transferred along a food chain or trophic level
carbon monoxide- colorless, odorless gas that at very low concentrations is extremely toxic to humans and animals
carbon dioxide- molecule of carbon and oxygen present in the atmosphere at approximately 350 ppm; emissions of carbon dioxide resulting from burning of fossil fuels are thought to be contributing to potential global warming through an enhanced greenhouse effect
chlorofluorocarbons (CFCs)- highly stable compounds that have been or are being used in spray cans as aerosol propellants and in refrigeration units, emissions of chlorofluorocarbons have been associated with potential global warming and stratospheric ozone depletion
deforestation- harvesting trees for commercial and other uses and burning forest to convert lands to agricultural purposes
El Ni�o- natural perturbation of the physical earth system that affects global climate; characterized by development of warm oceanic waters on the eastern part of the tropical Pacific Ocean, a weakening or reversal of trade winds, and a weakening or even reversal of the equatorial ocean currents; reoccurs periodically and affects the atmosphere and global temperature by pumping heat into the atmosphere
Environmental Protection Agency (EPA)- an organization set up to monitor the use of pollutants and conserve the environment
hydrochlorofluorocarbons- contain an atom of hydrogen in place of a chlorine and may be broken down in lower atmosphere and thus not inject chlorine into the stratosphere
hydrofluorocarbons- do not contain chlorine, but do contain fluorine and when fluorine atoms are released into the stratosphere they participate in reactions similar to those of chlorine and can cause oxygen depletion
methyl bromide- broad spectrum pesticide used in the control of pest insects, nematodes, weeds, pathogens, and rodents; used primarily for soil fumigation effectively sterilizing the soil, killing the vast majority of soil organisms
mitigation- process that identifies actions to avoid, lessen, or compensate for anticipated adverse environmental impacts
Montreal Protocol, The- landmark international agreement designed to protect the stratospheric ozone layer; originally signed in 1987 and substantially amended in 1990 and 1992; stipulates that the production and consumption of compounds that deplete ozone in the stratosphere are to be phased out by 2000
ozone- form of oxygen in which three atoms of oxygen occur together; is chemically active and had a short average lifetime in the atmosphere; forms a natural layer high in the atmosphere that protects us from harmful UV radiation from the sun; is an air pollutant when present in the lower atmosphere above the National Air Quality Standards
ozone depletion- when the ozone layer is chemically perturbed due to the presence of CFCs in the stratosphere
particulate matter- small particles of solid or liquid substances that are released into the atmosphere by many activities, including farming, volcanic eruption, and burning fossil fuels; particulates affect human health, ecosystems, and the biosphere
radioisotopes- affect the environment by emitting radiation that affects other materials and by entering the normal pathways of mineral cycling and ecological food chains
radon- naturally occurring radioactive gas; radon is colorless, odorless, and tasteless and must be identified through proper testing
stratosphere- the region of the upper atmosphere extending from the tropopause (8 to 15 km altitude) to about 50 km; the thermal structure is determined by its radiation balance and is generally very stable with low humidity
subaqueous- existing, formed, or taking place in or under water
ultraviolet (UV) radiation- consists of wavelengths between 0.1 �m and 0.4 �m and is further subdivided into UVA, UVB, and UVC; potentially damaging to life on earth
References
World Wide Web Sites Used:
A Range of Initiatives
Climate Change: Some Basics
Climate Observations Substantiate Global Warming
Frequently Asked Questions on the Methyl Bromide Phase Out
Glossary on Ozone Depletion
Introduction to Atmospheric Chemistry
Questions and Answers on Ozone Depletion
The Montreal Protocol on Substances that Deplete the Ozone Layer
What is Sustainable Agriculture?
Why Does Atmospheric CO2 Rise?
Encyclopedias and Books Used:
"Air Pollution," Microsoft Encarta ® 96 Encyclopedia. 1996.
Botkin, Daniel, and Keller, Edward, 1995 Environmental Science (John Wiley & Sons)
Broecker, Wallace S., 1985 How to Build a Habitable Planet (Wallace S. Broecker)
Charlier, R.H., and Justus, J.R. 1993 Ocean Energies (Elsevier Science Publishers)
Commoner, Barry 1992 Making Peace With the Planet (The New Press)
Holdren, John, and Herrera, Philip 1971 Energy (the Sierra Club)