P.F.S.R.L. - AP Mutants

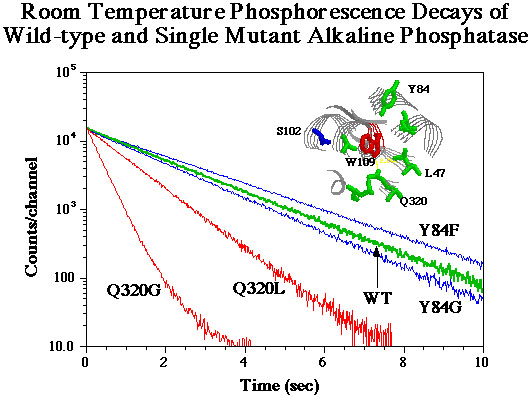
Displayed above is raw data collected from
our PMT during Phosphorescence experiments with WT-AP and several mutants.
The different slopes to the decay curves indicate different RTP lifetimes.
The insert shows tryptophan 109 and the local of the amino acids
mutated in our lab.
In order to test theories about the
correlation between local environmental effects and the RTP lifetime of
tryptophan residues (and more generally about the correlation between
internal structure and global stability) we made several amino acid
mutations within the core of AP near the phosphorescing Tryptophan (W).
Tryosine (Y) amino acids are known
quenchers of phosphorescence of NATA in solution6
and Y84 is very close to W109 in AP. Changing this Tyrosine to a
Phenylalanine (F) residue, which is roughly the same size as Tyrosine but
not as strong a quencher increased the RTP lifetime (the Y84F mutant)
relative to the wild type (WT) protein. If the Tyrosine was replaced with
a Glycine (G), which is not an effective quencher, but is much smaller
that either Tyrosine or Phenylalanine, the RTP lifetime (the Y84G mutant)
is decreased relative to WT. This can be attributed to a decrease in
the local rigidity of W109's environment because of the increased size
of the cavity caused by the small glycine.
The Glutamine residue (G) at position 320
is thought to hydrogen bond to W109 and therefore stabilize it. This
stabilization is though to increase the RTP lifetime by making the
environment of W109 more rigid. If this residue is replaced by a
Leucine (L), which is roughly the same size as Glutamine but can't
hydrogen bond to W109, the RTP lifetime is decreased compared to WT.
If this residue is replace by a Glycine the lifetime drops even further,
which can, as before, be attributed to an increase in the cavity size
around W109.
While changes in the RTP lifetime can be
drastically altered by these mutations, enzyme activity and thermodynamic
stability of the holo enzymes are less affected.

Main Page
Current Research
AP Research


![]()