Objective: Determine if resin composites containing bioactive glass fillers are capable of releasing fluoride and being recharged with fluoride.
Methods: Composites were produced with 50:50 BisGMA:TEGDMA, 0.4% CQ, 0.8% EDMAB, and 0.05% BHT, combined with spherical 0.4 µm Sr 35 filler (Bisco) mixed with 5, 10, or 15wt% F-containing bioactive glass (BAG) to 70wt% total filler. The BAG contained (mole %) 62Si, 1B, 3F, 4P, and 30Ca. Control composites of identical composition contained aerosol silica filler (OX-50, Degussa) instead of BAG. Composites (4 mm diameter x 1 mm thick) were light-cured (Demi; Kerr: 573 mW/cm2) for 40 s and stored dry for 24 h. Disks (n = 5) were then immersed in 2 mL DI water for 2, 20, and 200 h. The disks were removed after each time point, rinsed with DI water, and re-immersed in fresh DI water. Solutions (1 mL) at each time were combined with low-level TISAB (1 mL) and fluoride concentrations measured with a fluoride ion specific electrode (Orion 9409BN). After 200 h, the disks were lightly abraded, rinsed and re-immersed in fresh DI water. Fluoride concentrations were re-measured after 2 h and 20 h. The disks were then placed in a 5,000 ppm NaF fluoride solution for 5 min, rinsed and re-immersed in DI water. Fluoride was re-measured after 2, 20, and 200 h. Fluoride levels were compared by 2-way ANOVA/Tukey’s(=0.05).
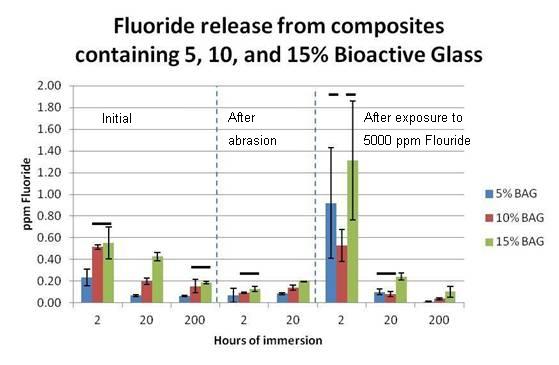
Results: Fluoride continued to be released (15% 10% > 5% BAG-composite) up to 222h of immersion (Figure; connected bars not significantly different). Abrasion did not increase fluoride release, but all composites showed significant increases in fluoride release after exposure to 5,000ppm F.
Conclusion: The addition of fluoride-containing bioactive glass to a composite serves as both a reservoir for fluoride release as well as a material that can be readily recharged with fluoride.
Keywords: Bioactive Glass, Composites and Fluoride
![[ Visit AADR's Website ]](images/banner.jpg)