Observation-based methods
Observation-based methods (OBMs) for analyzing urban/regional ozone production and Ozone-NOx-VOC sensitivity.
Dr. Sanford Sillman
Research Scientist
University of Michigan
sillman@umich.edu
Access and download this document as PDF.
Acknowledgement: This site was prepared in conjunction with research supported by the U.S. Environmental Protection Agency under the Science To Improve Results (STAR) program (grant #R826765) and by the EPA Office of Research and Development (grant #F005300).
Assistance was also provided by the National Science Foundation (grant
#ATM-0207841
) and by the
Southern Oxidants Study
.
Although the site was prepared with the aid of funding from EPA, it has not been subjected to peer and administrative review by either agency, and therefore may not necessarily reflect the views of the agency, and no official endorsement should be inferred.
Observation-based methods (OBMs) refer to attempts to evaluate the sources of atmospheric ozone in urban and polluted rural environments based on inferences made directly from measurements. They are also closely related to the question of how ozone is related to its two main precursors: nitrogen oxides (NOx) and volatile organic compounds (VOC).
This site is intended to provide a guide for researchers and policymakers who would like to use OBMs as part of their evaluation of the sources of ozone. The methods and results shown here are the authors' recommendations only and do not have official support from the U.S. EPA or any other agency.
Complete results:
A
draft report to EPA is available here as a PDF.
Also at this site:
[Top of page]
1. Observation-based methods - Overview. [Go to Section 1]
2. Secondary species as NOx-VOC indicators [Go to Section 2]
4. Methods based on ambient NOx and VOC [Go to Section 4]
[Return to Table of Contents]
The following gives a concise summary of each of the methods included here. It also introduces the detailed presentation for each OBM and the contents of each.
Contents
The primary purpose of OBMs is to reduce the uncertainty in predictions for the impact of reduced NOx and VOC on O3 by using ambient measurements. O3-NOx-VOC sensitivity is a major source of uncertainty in ozone-precursor predictions (See overview of
ozone, Figure 1.3)
A second purpose is to evaluate the accuracy of chemistry/transport models , which seek to represent the process of ozone formation and transport and often provide the basis for predicting the impact of emissions on O3.
A third purpose is to evaluate the accuracy of emission inventories , which often represent the major uncertainty in chemistry/transport models.
They also provide information about the ozone production efficiency (OPE) per NOx , which represents the ratio of ozone production to NOx removal.
OBMs often provide methods to infer whether ambient O3 is NOx-sensitive or VOC-sensitive based on ambient measurements. These may include simple rules-of-thumb that identify conditions as NOx-sensitive or VOC-sensitive based on the value of certain measured indices. Here, caution is recommended in the use of simple rules of thumb. It is more reliable if the OBM can be expanded to include a broad analysis of measured data sets , which would evaluate the accuracy and applicability of rules of thumb and would identify errors in measurements. This analysis represents essential quality assurance for the OBM.
The following gives an overview of individual methods. Each method is linked to a detailed presentation.
[Return to Table of Contents]
[Top of section]
[Go to detailed presentation]
This approach uses measured secondary species - primarily reactive nitrogen and peroxides - to derive information about sensitivity. The sum of total reactive nitrogen (NOy, including primary NOx) is also used.
The secondary species have little direct impact on the ozone formation process. They are usually produced simultaneously with ozone and thus provide information about the conditions under which ozone was formed.
NOx-VOC indicators are based on results from chemistry/transport models. The model results predict that certain values of NOx-VOC indicators are associated with NOx-sensitive conditions, while other values are associated with VOC-sensitive conditions. Some confirmation is provided by results from ambient measurements, which are consistent with predictions associated with indicators.
Initially, the indicator approach was presented as a series of rules-of-thumb that would identify whether Ox was NOx-sensitive or VOC-sensitive based on ambient measurements. Ratios such as (where NOz represents summed NOx reaction products, or NOy-NOx), and O3/HNO3and H2O2/HNO3 were identified as indicators for NOx-sensitive chemistry if they exceeded a certain threshold or transition value, and as indicators for VOC-sensitive chemistry if they fell below that value (Sillman, 1995). Subsequent results suggested that the behavior of indicator ratios may be different in different locations (Lu and Chang, 1998).
An expanded version of the indicator concept uses measured patterns of correlation between secondary species (e.g. O3 vs. NOz, etc.) to make inferences about NOx-VOC sensitivity and to evaluate chemistry/transport models. This approach is advantageous because the measured correlation patterns can be used to identify erroneous measurements or to identify inconsistencies that would prevent the use of indicator ratios to infer NOx-VOC sensitivity.
The measured correlation patterns provide a method for evaluating chemistry/transport models. Because the indicator correlations are related to NOx-VOC sensitivity in models, they provide an test for the accuracy of model NOx-VOC sensitivity predictions.
The detailed presentation below provides the following.
The "rules of thumb" for indicator ratios.
Indicator correlation patterns for NOx-sensitive and VOC-sensitive locations, from chemistry/transport models and from measurements.
Use of indicator correlations to evaluate chemistry/transport models.
Guidelines for practical implementation.
[Top of section]
[Go to detailed presentation]
Smog production algorithms represent an attempt to identify NOx-sensitive and VOC-sensitive conditions based on ambient measurements of O3, NOx and/or NOy.
The algorithms consist of "rules of thumb" for NOx-sensitive versus VOC-sensitive conditions, derived based on the results of smog chamber experiments.
The "extent-of-reaction" concept posits that NOx-sensitive conditions are found when photochemistry has been run to completion and most of the emitted NOx has been removed, while VOC-sensitive conditions are found when the ozone production process has not yet run to completion.
Smog production algorithms have been widely used by EPA and by state governments to evaluate ozone-precursor relationships. They are not recommended here. As presented here, the smog production algorithms contain fundamental flaws that should preclude their use. The detailed presentation [Link, Section 3] below identifies these flaws.
For a more positive view of smog production algorithms, refer to Blanchard et al. (1999, 2000) and Blanchard and Stockenius (2001). A more comprehensive description is included in the
draft report to EPA.
[Top of section]
[Go to detailed presentation]
NOx and VOC are direct precursors of ozone formation, and are directly related to O3-NOx-VOC sensitivity.
Ambient NOx and VOC are related to the instantaneous rate of ozone production. It is more difficult to relate ambient NOx and VOC to ambient O3, because transport and upwind production must be accounted for.
It is possible to calculate the instantaneous rate of ozone production and its sensitivity to NOx and VOC, using a 0-d model and measured NOx and VOC. Kleinman et al. (1997, 2000, 2001,2005) and Tonnessen and Dennis (2000) have also developed simplified formulas for instantaneous NOx-VOC sensitivity, based on ambient NOx and reactivity-weighted VOC.
Cardelino et al. (1995, 2000) built a model to calculate overall NOx-VOC sensitivity in an urban area, based on calculated rates of ozone production at measurement sites throughout the area, driven by measured ambient NOx and VOC.
Analysis based on ambient NOx and VOC form a natural complement to the analysis based on secondary species as NOx-VOC indicators.
Methods for calculating ozone production rates and NOx-VOC sensitivity should be combined with a broader evaluation of measurements, including measured correlations between individual VOC and between VOC and NOx, inferred emission rates in comparison with inventories, and quality assurance. These methods are described in the next section (1.5).
The detailed presentation includes a combined description of methods for deriving NOx-VOC sensitivity from ambient NOx and VOC, methods for deriving emission rates, and the recommended broader analysis of measurements.
Update: Duncan et al (2010) describe space-based measurements of HCHO and NO2 and use the ratio HCHO/NO2 as a diagnostic for O3-NOx-VOC sensitivity.
[Top of section]
[Go to detailed presentation]
Emission inventories represent the single largest uncertainty in chemistry/transport models for ozone.
Emission inventories can be evaluated using data sets of measured ambient NOx (or NOy) and speciated VOC. Parrish et al. (1998, 2001) have described methods for inferring emission rates based on correlations between individual VOC and between VOC and NOx. These methods also include tests for internal consistency of measurements.
The measured VOC-VOC and VOC-NOx correlations and/or directly inferred emission rates can be used to evaluate the accuracy of chemistry/transport model applications, and to modify emission rates in these models to insure better agreement with ambient measurements.
Mendoza-Dominguez and Russell (2001a, 2001b) have used measured NOx and VOC in combination with chemistry/transport models to modify emission inventories, using techniques of inverse modeling.
Evaluation of ambient NOx and VOC and consistency tests can be combined with analysis of NOx-VOC sensitivity based on ambient NOx and VOC.
The detailed presentation for ambient NOx and VOC includes the following:
- Methods for inferring NOx-VOC sensitivity from ambient NOx and VOC.
- Methods for inferring emission rates from ambient NOx and VOC.
- Tests for consistency of the data set for NOx and VOC.
- Suggestions for practical implementation.
[Return to Table of Contents]
CONTENTS:
[Top of section].
NOx-VOC indicators mainly involve secondary species that are produced concurrently with photochemical production of O3. These species are all relatively long-lived and are transported along with O3. Ratios among these species carry information about the chemistry associated with ozone formation.
Peroxides and nitric acid (HNO3) are directly related to the chemistry that causes the split into NOx-sensitive and VOC-sensitive regimes. Under NOx-sensitive conditions the rate of production of peroxides exceeds the rate of production for HNO3. Under NOx-saturated (VOC-sensitive) conditions the rate of production of HNO3 exceeds the rate of production of peroxides. These production rates are determined by the chemistry of odd hydrogen radicals (OH, HO2, CH3O2, etc.), which also determine O3-NOx-VOC sensitivity. (A detailed description of the chemistry of O3, NOx and VOC is available at this site. [Link coming in future]).
The ambient ratio of peroxides to HNO3 reflects the rates of production of these species, except during conditions with rapid removal of these species (rainfall, or dry deposition from a shallow surface layer at night).
Ozone and NOx reaction products (NOz) are also related to the NOx-VOC chemistry, although the relation is less direct.
The ratio of rate of production of O3 to the rate of production of nitric acid (p(O3)/p(HNO3) is closely associated with the ratio of reactivity-weighted VOC to NOx. This is because production of O3 is initiated by the reaction of VOC with OH, while the production of HNO3 occurs through the reaction of NO2 with OH. The ratio (p(O3)/p(HNO3) is also related to the chemistry of odd hydrogen radicals which determines the split into NOx-sensitive and VOC-sensitive regimes (see description of chemistry of ozone [Link coming in the future]).
The ratio of production of O3 to removal of NOx (through chemical conversion to HNO3 and organic nitrates) is also related to the ozone production efficiency per NOx (OPE), which is often used in analyses of ozone production chemistry (e.g. Liu et al., 1987, Lin et al., 1988, Trainer et al., 1993).
The slope of ambient O3 versus summed NOx reaction products (NOz) is not exactly equal to the ozone production efficiency, because the slope is also affected by processes that remove NOx reaction products from the atmosphere (Sillman et al., 1998). However, variations in the slope of O3 versus NOz and O3 versus HNO3 reflect variations in both ozone production efficiency and O3-NOx-VOC sensitivity.
The ratio between ozone and organic nitrates is completely unrelated to O3-NOx-VOC sensitivity. The ratio of ozone to NOz carries information about O3-NOx-VOC sensitivity only because HNO3 is a major component of NOz.
Ozone and total reactive nitrogen (NOy): The ratio and correlation between O3 and NOy reflects two separate processes, both of which are related to O3-NOx-VOC sensitivity. It reflects the process of photochemical production of O3, represented by the ratio between O3 and NOz. It also reflects the process of NOx titration, which refers to the immediate removal of O3 through reaction with directly emitted NO. The other secondary species are unaffected by NOx titration. A low value for O3/NO3 identifies NO3 saturated chemistry either in the past history of the air mass (reflected by O3 versus NOz) or in current conditions (reflected by O3 versus NOx).
[Top of section].
The relation between O3-NOx-VOC sensitivity and NOx-VOC indicators was derived from chemistry-transport models. It was found that when models predict VOC-sensitive conditions, they also predict low values for certain ambient indicator ratios. When models predict NOx-sensitive conditions, they predict high values for the ratios. The same relation between predicted NOx-VOC sensitivity and indicator ratios appeared in models for several urban areas and regions in the U.S. and in Europe (Sillman, 1995; Sillman et al., 1997, 1998, 2002; Sillman and He, 2002; Tonnessen et al., 2000b; Martilli et al., 2002).
The indicator ratios identified in the above references are: O3/NOy, O3/NOz, O3/HNO3, H2O2/HNO3, total peroxides/HNO3, and equivalent ratios of peroxides to NOz and NOx. Ratios were also identified that used background values of ozone (O3b) and other species: (O3-O3b)/(NOy-NOyb), (O3-O3b)/(NOz-NOzb), and (O3-O3b)/(HNO3-HNO3b). This is based on a specific definition of background ozone, given below. These ratios refer to values between the hours of noon and sunset only, and are related to O3-NOx-VOC sensitivity only at the same time and location as the ambient ratio.
Figure 2.1 shows the relation between NOx-VOC sensitivity in models. The figure shows the predicted reduction in O3 resulting from either a 35% reduction in emissions of anthropogenic VOC or a 35% reduction in NOx, for models for several regions in the U.S. (listed in Table 2.1). Each point in the figure represents a different model location. All are for afternoon hours, usually close to the time of maximum O3. The split between NOx-sensitive and VOC-sensitive locations is clearly visible in the figures. (See description of O3-NOx-VOC elsewhere in this site [Link coming]). It can be seen that the primarily NOx-sensitive model locations also have high values for indicator ratios, and VOC-sensitive locations have low values for indicator ratios.
Indicator values for NOx-sensitive and VOC-sensitive conditions are summarized in Table 2.2 . Measured values lower than the NOx-VOC "transition" in the table are generally VOC-sensitive. Measured values higher than the NOx-VOC transition are generally VOC-sensitive. The median values identify typical indicator ratios for strongly NOx-sensitive and strongly VOC-sensitive conditions.
Definition: Background values for O3 and other species are defined as the ambient mixing ratios at a relatively unpolluted upwind site, as measured (or modeled) at as close as possible to the same time as the other ambient values (O3, NOy, etc.) of the indicator ratio. This nonstandard definition is used because with this definition it is easy to determine background values from a network of ambient measurements. Model results for indicator ratios were derived based on the same definition. Background values are based on measurements (or model values) at the same time as the other mixing ratios (typically during the afternoon hours) in order to include the impact of entrainment from aloft as the convective mixed layer grows. Given a network of measurements, background values should be selected based on the site with the lowest NOy for the hour of interest.
WARNING: Somewhat contrary results have been reported by Lu and Chang (1998), Chang et al. (1999) and Blanchard and Stockenius (2000).
It is more accurate to include an examination of measured correlations between secondary species rather than simple rules of thumb. Measured correlation patterns can be compared with predicted correlation patterns from models with NOx-sensitive and VOC-sensitive chemistry and with previous measurements. Indicator measurements provide valid information about O3-NOx-VOC sensitivity only if they show a correlation pattern that is consistent with results from either NOx-sensitive or VOC-sensitive models. If measurements fail to show this agreement, than the ratios are not valid as NOx-VOC indictors. This is described below.
720x720, plus filter/sharpen>
.jpg)
Figure 2.1 (first part).
Predicted reductions in ozone in response to a percent reduction in emissions of anthropogenic VOC (crosses), and predicted reductions in response to the same percent reduction in emissions of anthropogenic NOxg (green circles), plotted versus model values for proposed indicator ratios: O3/NOy, O3/NOz, O3/HNO3, and (H2O2+ROOH) /HNO3. Results are shown for four separate model scenarios (Lake Michigan, northeast, Nashville, and Los Angeles, from Table 2.1). Percent reductions are either 25% or 35% in individual scenarios.
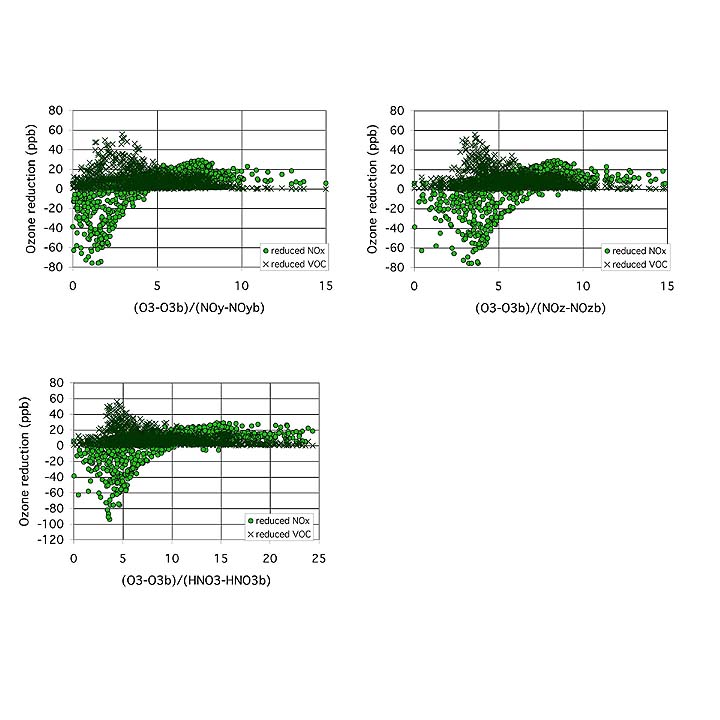
Figure 2.1 (continued). Predicted reductions in ozone in response to a percent reduction in emissions of anthropogenic VOC (crosses), and predicted reductions in response to the same percent reduction in emissions of anthropogenic NOxg (green circles), plotted versus model values for proposed indicator ratios: O3/NOy, O3/NOz, O3/HNO3, and (H2O2+ROOH) /HNO3. Results are shown for four separate model scenarios (Lake Michigan, northeast, Nashville, and Los Angeles, from Table 2.1). Percent reductions are either 25% or 35% in individual scenarios.
Table 2.1:
3-d simulations used in Figure 2.1
|
Location
|
Model
|
Photochemistry
|
Model Domain
|
Comparison w/measurements
|
Reference
|
|
Nashville
|
Sillman et al., 1998
| modified Lurmann et al., 1986
|
5x5 km urban; upwind domains includes eastern U.S.
|
O3, NOy, peroxides
| Sillman et al., 1998
|
|
Lake Michigan
|
Sillman et al., 1993
|
modified Lurmann et al., 1986
|
20x20 km in region; upwind domains includes eastern U.S
|
O3
|
Sillman, 1995
|
|
Northeast corridor
|
Sillman et al., 1993
|
modified Lurmann et al., 1986
|
20x20 km in region; upwind domains includes eastern U.S
|
O3
|
Sillman, 1995
|
|
Atlanta
|
UAM-IV, Morris and Myers, 1990
|
CB4 (Gery et al., 1989)
|
5x5 km urban
|
O3, NOy, isoprene, HCHO, other VOC
|
Sillman et al., 1997
|
|
San Joaquin (Sillman)
|
MAQSIP (Odman and Ingram, 1996)
|
CB4 (Gery et al., 1989) 12x12 km; domain includes all central California
|
O3
|
Sillman et al., 2001
|
|
Los Angeles (Godowitch)
|
UAM-IV, Morris and Myers, 1990
|
CB4 (Gery et al., 1989)
|
5x5 km urban
|
O3, NOy, NOz
|
Godowitch et al., 1994; Sillman et al., 1997
|
Table 2.2.Values of indicator ratios for NOx-sensitive, transitional, and VOC-sensitive conditions
The VOC-sensitive and NOx-sensitive values represent typical values for strongly VOC-sensitive and strongly NOx--sensitive locations, derived from chemistry/transport models. Ratios lower than the transition values generally correspond to VOC-sensitive conditions. Ratios higher than the transition values generally correspond to NOx-sensitive conditions.
|
indicator
|
median VOC-sensitive
|
transition
|
median NOx-sensitive
|
|
O3/NOy
|
5
|
6-8
|
11
|
|
O3/NOz
|
6
|
8-10
|
14
|
|
O3/HNO3
|
9
|
12-15
|
20
|
|
(O3-O3b)/(NOy-NOyb)
|
3
|
3.5-5
|
6
|
|
(O3-O3b)/(NOz-NOzb)
|
4
|
5-6
|
7
|
|
(O3-O3b)/(HNO3-HNO3b)
|
5
|
7-10
|
12
|
|
H2O2/HNO2
|
.15
|
.25-.35
|
.6
|
|
total peroxides/HNO3
|
.2
|
.2-.5.
|
9
|
|
H2O2/NOz
|
.12
|
.2-.25
|
.4
|
|
total peroxides/NOz
|
.15
|
.25-.35
|
.7
|
|
H2O2/NOy
|
.08
|
.12-.17
|
.35
|
|
total peroxides/NOy
|
.12
|
.20-.25
|
.6
|
[Top of section].
Models: Chemistry-transport models predict distinct patterns of correlation between O3 and NOy, between O3 and NOz, and between O3 and HNO3. These patterns are different for NO3-sensitive and VOC-sensitive conditions.
Figure 2.2 shows O3 vs. NOy, O3 vs. NOz, and O3 vs. HNO3 from simulations for several cities in the U.S. (listed in Table 2.1), afternoon hours only. The results are sorted based on predicted O3-NOx-VOC sensitivity at the time and location corresponding to the ambient value. They are sorted as NOx-sensitive, VOC-sensitive, mixed, or dominated by NOx titration (see Definition below).
NOx-sensitive locations all show a strong correlation between O3 and NOy, between O3 and NOz, and between O3 and HNO3, with a steep slope.
VOC-sensitive locations also show a correlation between O3 and NOz, and between O3 and HNO3, but the range of values is different and the slope is lower. VOC-sensitive locations may also have a positive correlation between O3 and NOy or they may have little or no correlation between O3 and NOy.
This figure is also available as a
data file (click here).
Figure 2.3 shows correlation patterns for O3 vs. NOy in individual simulations. These may look different from the composite correlation pattern. It is important to recognize that even a VOC-sensitive simulation may have a strong correlation between O3 and NOy (e.g. Lake Michigan).
2.3a. Definition: NOx-VOC sensitivity:
Locations are defined as NOx-sensitive if a percent (25%-50%) reduction in NOx emissions would cause a significant (>5 ppb) reduction in O3, and if the resulting O3 is significantly lower than would result from the same percent reduction in anthropogenic VOC.
Locations are defined as VOC-sensitive if a percent reduction in VOC would cause a significant reduction in O3, and if the resulting O3 is significantly lower than would result from the same percent reduction in NOx.
Locations are defined as mixed if a percent reduction in NOx would not result in O3 that is significantly higher or significantly lower than the same percent reduction in VOC.
Locations are defined as dominated by NOx titration if a percent reduction in NOx would cause a significant increase in O3, while a percent reduction in VOC would not cause a significant reduction in O3.
.jpg)
Figure 2.2a. Correlations for (a) O3 vs. NOy (in ppb) from the 3-d simulations listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. From Sillman and He (2002). This figure is also available as a
data file .
.jpg)
Figure 2.2b. Correlations for (b) O3 vs. NOz (in ppb) from the 3-d simulations listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. From Sillman and He (2002). This figure is also available as a
data file .
.jpg)
Figure 2.2c. Correlations for (c) O3 vs. HNO3 (in ppb) from the 3-d simulations listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. From Sillman and He (2002). This figure is also available as a
data file .
.jpg)
Figure 2.3. Correlations for O3 vs. NOy in ppb from individual 3-d
simulations listed in Table 2.1. Each location is classified as NOx-sensitive
(green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity
(lavender squares), and dominated by NOx titration (blue asterisks) based on
definitions in the text. Models are (a) Los Angeles (Godowitch),
(b) Lake Michigan, (c) northeast corridor, (d) Nashville, (e) Atlanta,
and (f) San Joaquin (Lu and Chang).
Measurements for O3 vs. NOz show a range of correlation patterns that is similar to model values. These are shown in Figure 2.4.
.jpg)
Figure 2.4. Measured correlations between O3 and NOz, both in ppb. Measurements are shown from field campaigns in Nashville (pink dashes), Paris (blue diamonds), Los Angeles (X's) and from four rural sites in the eastern U.S. (green circles). From measurements reported by Sillman et al., 1997, 1998, 2002 and Trainer et al., 1993.
[Top of section].
Model-measurement comparisons for ozone and reactive nitrogen provide a way to evaluate the accuracy of models and also to evaluate whether measurements can properly be interpreted as evidence for NOx-VOC sensitivity.
The two critical evaluations are: (i) full-domain correlations between O3 and NOz, etc. (afternoon values only); and (ii) indicator species values associated with peak and near-peak O3.
Here are examples.
A primarily NOx-sensitive model: Nashville (Figure 2.5). Model and measured O3 vs. NOz both show a strong positive correlation. The range of values of O3 and NOz are similar in the model and in measurements. Peak O3 and NOz associated with peak O3 in the model differ from the measured peak O3 and associated NOz by 10% or less, suggesting good model-measurement agreement. The ratio O3/NOz associated with peak O3 is lower in the model than in measurements by 15% (8.4 model, 9.7 measurements). This difference is too small to suggest bias in model NOx-VOC sensitivity predictions.
.jpg)
Figure 2.5. Measured correlation between O3 and NOz (ppb) (blue diamonds), compared with model results for Nashville (Sillman et al., 1998). Each model location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text.
A VOC-sensitive model: Los Angeles (Figure 6).
The model scenario shows a positive correlation between O3 and NOz, but with scatter, and with mostly VOC-sensitive chemistry. Measurements also show a positive correlation with scatter, and with a similar range of O3 vs. NOz, The measurements correspond closely to model values that are associated with VOC-sensitive conditions. The model also shows some NOx-sensitive locations, but very few measurements correspond to the NOx-sensitive O3 vs. NOz in the model. The model predicts peak O3 with both mixed and VOC-sensitive conditions. Peak O3 is underestimated by 10% vs. measurements, but NOz and O3/NOz are both underestimated by approximately 30%. The difference between model and measured O3/NOz at peak O3 (7.9 model, 6.0 measured) corresponds to the difference between somewhat VOC-sensitive conditions and strongly VOC-sensitive conditions in Figure 2.1.
This suggests that the model scenario, though primarily VOC-sensitive, may still be somewhat biased towards NOx-sensitive chemistry.
.jpg)
Figure 2.6. Measured correlation between O3 and NOz (ppb) (blue diamonds), compared with model results for Los Angeles (Sillman et al., 1997). Each model location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text.
A biased model: Atlanta (Figure 7): The first model scenario (Figure 7a) predicts VOC-sensitive conditions associated with peak O3 in the Atlanta urban plume and NOx-sensitive conditions elsewhere. Measured O3 vs. NO3 agrees with the model values for NOx-sensitive regions of the model, but disagree with model values for VOC-sensitive regions only. Model peak O3 is in good agreement with measured peak O3, but NOy and O3/NOy in the vicinity of peak O3 are both higher in the model than in measurements by nearly a factor of two.
A modified model scenario with strongly NOx-sensitive conditions (Figure 5b) shows better agreement with measurements. Model peak O3, NOy and O3/NOy in the vicinity of peak O3, all are within 15% of measured values.
In this evaluation, the VOC-sensitive and NOx-sensitive model scenarios both show good agreement with measured O3. An evaluation using O3 vs. NOy is necessary to identify model errors.
.jpg)
(a)
.jpg)
(b)
Figure 2.7. Measured correlation between O3 and NOz (ppb) (blue diamonds), compared with model results for Atlanta (Sillman et al., 1997). Each model location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text.
An event where indicators are invalid: (unpublished data, Figure 2.8): Here, measured O3 vs. NOz differs from model values for the whole model domain. The measured O3 vs. NOz does not correspond to model results for either NOx-sensitive or VOC-sensitive cases. This type of model-measurement discrepancy might be caused by one of several factors: loss of reactive nitrogen through wet deposition or through aerosol interactions, unknown photochemical processes, or erroneous measurements. Based on this model-measurement discrepancy, it is not valid to draw inferences about Ox-NOx-VOC sensitivity from the measurements.
.jpg)
Figure 2.8. Measured correlation between O3 and NOz (ppb) (blue diamonds), compared with model results for Paris (unpublished). Each model location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text.
[Top of section].
2.5.1. Ozone vs organic nitrates.
The correlation between O3 and organic nitrates is completely unrelated to O3-NOx-VOC sensitivity. As shown in Figure 2.9, the correlation between O3 and organic nitrates is the same for both NOx-sensitive and VOC-sensitive conditions.
.jpg)
Figure 2.9. Correlations for organic nitrates (defined as NOy-NOx-HNO3, in ppb) from the 3-d simulations listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text.
2.5.2. Peroxides:
If measured H2O2 and/or organic peroxides are available, a much stronger evaluation of model accuracy can be provided.
Models predict that peroxides vs. HNO3 show a very different range of values for NOx-sensitive and VOC-sensitive conditions. (Figure 2.10).
There is no predicted correlation between peroxides and HNO3 or between peroxides and NOz. However, models predict a strong, consistent correlation between O3 and the sum 2H2O2+NOz (Figure 2.11). This correlation is similar for both NOx-sensitive and VOC-sensitive conditions and provides a test for general validity of indicator ratios.
When measurements agree with model values for this correlation, it guarantees that the indicator ratios O3/NOz and 2H2O2/NOz are consistent with each other - both ratios will suggest the same O3-NOx-VOC sensitivity. Errors that would invalidate indicator ratios (e.g. removal of HNO3 through wet deposition or aerosol interactions) would also cause a model-measurement discrepancy for O3 vs. 2H2O2+NOz.
.jpg)
Figure 2.10. Correlations for total peroxides vs. HNO3 ( in ppb) from the 3-d simulations listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. From Sillman and He (2002).
.jpg)
Figure 2.11. Measured correlation between O3 and the sum 2H2O2+NOz (ppb) in Nashville (blue diamonds), compared with model results. Each model location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. The model and measurements are from Sillman et al. (1998).
[Top of section].
Use of indicator ratios to evaluate O3-NOx-VOC sensitivity requires a network of measured O3 and either NOy or HNO3. The value NOz is derived from measured NOy and NOx.
The network must be extensive enough to include the region with the likely peak O3. Measured secondary species only would provide information about conditions at the measurement site.
The measured total reactive nitrogen (NOy) must include gas-phase HNO3. It is not possible to substitute NOx or the sum of NOx and organic nitrates (supplied by some types of instruments associated with the PAMS network) for NOy. As described elsewhere, the connection between NOy and O3-NO3-VOC sensitivity is primarily due to HNO3 rather than to other components of NOy. It is also important to insure that HNO3 is not lost in the inlet tubes of the NOy instrument. ((Luke et al., 1998, Parrish et al., 2000 and McClenny et al., 2000 provide information about this.)
Results should be evaluated separately on a day-to-day basis. Measurements affected by rainfall events (at the site or upwind) cannot be used as NOy-VOC indicators because HNO3 has been removed. This situations can sometimes be identified by analyzing measurements on a day-by-day basis.
Measured O3 versus NOy (or NOz or HNO3) should be plotted and tabulated for the afternoon hours only (12 noon to 2 hours before sunset). Values during the evening are likely to be affected by surface deposition, and different correlation patterns are expected during the morning.
The correlation between O3 and NOy, etc. should be superimposed on the pattern of model correlations for NOy-sensitive and VOC-sensitive conditions (Figure 2.2, also available as a
data file
) and/or measurements. To be valid, measured values should fall within the broad range of model values. If a significant portion of measurements fall outside the range of model values, then the measurements for the day in question should not be interpreted as NOy-VOC indicators.
Model-measurement comparisons for O3 and NOy, etc. should include both a general evaluation and an evaluation for peak and near-peak O3. This should be based on afternoon values only. They should include the complete ensemble of measurement sites within a metropolitan area, and use model ambient concentrations for the same times and locations of the measurements.
Model and measured ensemble correlations should be plotted for each day, as in Figures 2.5-2.8. It is useful to plot model correlations for both the entire model domain and for the measurement sites only, in order to identify whether the measurement sites are representative of conditions throughout the area of interest.
The following procedure is suggested:
-
Peak O3: Identify model NOy (or NOz or HNO3) and the ratio O3/NOy at the time and place of model peak O3, and measured NOy and O3/NOy, etc. at the time and place of measured peak O3.
- Near-peak O3: Identify the model mean NOy (or NOz or HNO3) and range of NOy values for times and locations with model O3 within 10% of the model peak value, and identify the measured mean NOy (or NOz or HNO3) and range of values for times and locations with measured O3 within 10% of the measured peak value,
- General correlation: Establish a series of intervals for the afternoon values of NOy, NOz or HNO3, based on the range of afternoon values in the model and in measurements: 0-2 ppb, 2-4, 4-6, 6-8, etc. Identify mean O3 for all model locations with NOy (or NOz or HNO3) in the specified interval and for all measurements with NOy in the specified interval. These values will be used for a model-measurement comparison.
Interpretation:
[Return to Table of Contents]
Smog production algorithms are not recommended for use here. Results shown here identify weaknesses in the method. For a more positive view, refer to Blanchard et al. (1999), Blanchard (2000) and Blanchard and Stockenius (2001).
The smog production algorithms consist of "extent of reaction parameters", which are calculated as function of either (i) ambient O3 and NOy, (ii) NOx and NOy, or (iii) Ox and NOx. A low extent of reaction (<0.6) suggests the presence of largely unprocessed direct emissions, and is interpreted as VOC-sensitive. A high extent of reaction (>0.9) suggests that photochemistry has been run to completion, and is interpreted as NOx-sensitive.
The following are weaknesses in the smog production algorithms.
Rule of thumb: The smog production algorithms consist only of rules of thumb, intended to tell whether ambient O3 is primarily sensitive to NOx or to VOC based on field measurements. They have never been linked to a broader analysis of ambient measurements that might evaluate the appropriateness of the method.
Reliance on smog chamber results: The smog production algorithms were derived empirically from results of smog chamber experiments. This is an advantage in one sense: the algorithms are not dependent on model calculations. However, they differ from ambient conditions in many ways.
- Smog chambers typically include a single rapid introduction of precursors, rather than continuous and varying precursor emissions as occurs in nature.
-
Smog chambers typically have VOC and NOx concentrations that are much higher than ambient concentrations at times of high ozone production.
-
Smog chambers do not include multi-day processes.
-
Dry deposition in smog chambers is very different from dry deposition outdoors.
Conceptual flaws: The central concept is that NOx-sensitive conditions are associated with a high "extent of reaction" - photochemically aged air that has had most of its NOx reacted away. VOC-sensitive conditions are associated with a low “extent of reaction”, including relatively fresh emissions.
Relatively fresh emissions are in fact more likely to have VOC-sensitive photochemistry, and aged downwind emissions are more likely to be NOx-sensitive (Milford et al., 1989, 1994). But this is not universally true, and there is no causal link.
Photochemically aged air can still be primarily VOC-sensitive if emission sources have low VOC/NOx ratios, low VOC reactivity, and little or no biogenic VOC. Ambient measurements have identified instances of photochemically aged air with apparently VOC-sensitive chemistry (Jacob et al., 1995, Hirsch et al., 1996, Kleinman et al., 2002). Such cases have also been found in chemistry-transport models. See, for example, the Lake Michigan case in Figure 2.3.
Relatively fresh emissions can be primarily NOx-sensitive if they have high VOC/NOx ratios and high VOC reactivity.
Results from chemistry/transport models: Figure 3.1 shows how predicted O3-NOx-VOC sensitivity compares with extent of reaction derived from predicted ambient conditions in chemistry/transport models. This is equivalent to Figure 2.1 for NOx-VOC indicators, with models listed in Table 2.1.
Three different extent-of-reaction parameters are used, as defined by Blanchard et al. (1999). They may be represented approximately as: (i) (O3-O3b)/19NOy0.67, (ii) (1-NOx/NOy)0.67, and (iii) (O3-O3b)/19NOx0.67. The complete formulas are given in the
draft report to EPA.
and in Blanchard et al., 1999.
The first extent parameter shows reasonably good agreement with O3-NOx-VOC sensitivity. The other parameters show worse agreement. VOC-sensitive conditions are found for almost the entire range of values of the second and third extent parameter.
The poor results are largely due to the simulation for Lake Michigan, because this simulation includes an aged urban plume from Chicago that remains VOC-sensitive as it travels downwind.
Figure 3.2 shows the same results in different format. Figure 3.2 also compares the extent parameter
(O3-O3b)/19NOy0.67 with a simpler alternative, (O3-O3b)/(NOy-NOyb). The simpler alternative (included above as a NOx-VOC indicator) gives better results.
.jpg)
Figure 3.1. Predicted reductions in ozone in response to a percent reduction in emissions of anthropogenic VOC (crosses), and predicted reductions in response to the same percent reduction in emissions of anthropogenic NOx (green circles), plotted versus model values for three extent parameters (B1, B2 and B3, defined as in Blanchard et al., 1999, and given in the
draft report to EPA.
), for the five model scenarios from Table 2.1. Percent reductions are either 25% or 35% in individual scenarios. Based on results shown in Sillman and He (2002).
.jpg)
(a) ExtentB1 (Equation 3.2.3)
.jpg)
(b) ExtentB2 (Equation 3.2.5)
Figure 3.2. Extent of reaction versus O3 for five model scenarios listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. Results are shown for the three extent parameters (B1, B2 and B3, defined in Equations 3.2.3, 3.2.5 and 3.2.8 in the
draft report to EPA.
from Blanchard et al., 1999). Results for the ratio (O3-O3b)/(NOy-NOyb) are shown for comparison. From Sillman and He (2002).
.jpg)
(c) ExtentB3 (Equation 3.2.8)
.jpg)
(d) (O3-O3b)/(NOy-NOyb)
Figure 3.2. Extent of reaction versus O3 for five model scenarios listed in Table 2.1. Each location is classified as NOx-sensitive (green circles), VOC-sensitive (crosses), mixed or with near-zero sensitivity (lavender squares), and dominated by NOx titration (blue asterisks) based on definitions in the text. Results are shown for the three extent parameters (B1, B2 and B3, defined in Equations 3.2.3, 3.2.5 and 3.2.8 in the
draft report to EPA.
from Blanchard et al., 1999). Results for the ratio (O3-O3b)/(NOy-NOyb) are shown for comparison. From Sillman and He (2002).
[Return to Table of Contents]
CONTENTS:
[Top of section].
Ambient NOx and VOC are directly related to the instantaneous rate of production of O3. Given measurements of NOx and primary hydrocarbons (along with meteorological data), it is possible to calculate the instantaneous rate of ozone production using a 0-d photochemical calculation.
Ambient NOx and VOC are also directly related to the NOx-VOC sensitivity of the instantaneous rate of ozone production. In particular, instantaneous NOx-VOC sensitivity is closely related to the ratio of reactivity-weighted VOC to NOx.
The ratio of reactivity-weighted VOC to NOx is also related to the ratio of production rates of ozone and HNO3, (p(O3)/p(HNO3), and to the ozone production efficiency . Because of this, the ambient slope between O3 and NOz or between O3 and HNO3 is linked to the ratio of reactivity-weighted VOC to NOx. Analyses based on ambient VOC and NOx and analyses based on secondary species must be consistent with each other.
There is no direct way to relate ambient NOx and VOC to NOx-VOC sensitivity for ambient O3 (as opposed to the instantaneous production rate of O3). Ambient O3 is affected chemistry and transport over an extensive upwind region. Ambient NOx and VOC only provide information about the instantaneous production rate at the time and place of measurements.
An old rule of thumb, relating O3-NOx-VOC sensitivity to the ambient VOC/NOx ratio in urban centers during the morning hours, is not correct. The old rule was based on a specific VOC speciation, omitted biogenic VOC, omitted multi-day transport, and omitted the complex geographical variation in emissions throughout a metropolitan area.
Tonnessen and Dennis (2000), Kirchner (2001) and Kleinman et al. (1997, 2000, 2001, 2002,2005) have developed simple rules of thumb that relate the NOx-VOC sensitivity of instantaneous production of O3 to ambient NOx and VOC. The rule developed by Tonnessen and Dennis (2000) and by Kirchner et al. (2001) are both equivalent to the following:
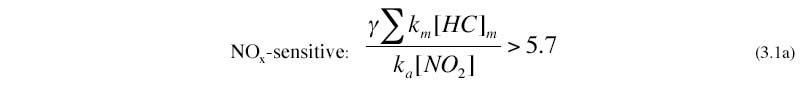
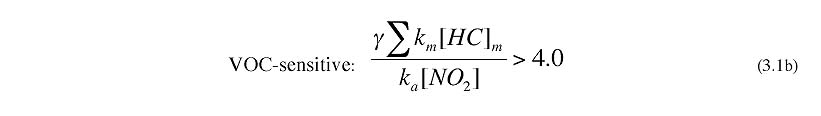
where km and ka are reaction rates for OH with individual hydrocarbons and with NO2, and GAMMA is an empirical correction factor (recommended as 1.3) to account for unmeasured hydrocarbons. The summation is performed over all primary and secondary hydrocarbons, and should also include oxygenated organics and CO.
(Measured VOC typically include only primary hydrocarbons and CO, with no oxygenated species. Guidance needs to be developed for the appropriate adjustment for this situation.)
Kleinman et al. (1997, 2000, 2001, 2002) developed a more complex rule that takes into account the variation in the radical source, which depends on ambient O3 and solar radiation. They have used their rule to analyze ozone production in New York, Philadelphia, Phoenix and Houston (results from all five cities in Kleinman et al., 2005). [These are available for download from: http://www.ecd.bnl.gov/publications.html]
Cardelino and Chameides (1995, 2000) developed a procedure for estimating overall O3-NOx-VOC sensitivity in a metropolitan area based on ambient NOx and VOC. Essentially, they calculate instantaneous production rates for O3 at all sites with measured NOx and VOC, and use the sum to represent ozone production within the metropolitan area. Their calculation also includes procedures for including unmeasured secondary organics.
Update: Duncan et al (2010) describe space-based measurements of HCHO and NO2 and use the ratio HCHO/NO2 as a diagnostic for O3-NOx-VOC sensitivity.
The above methods all require measurements of a relatively complete array of primary hydrocarbons, along with accurate measurements of either NO or NOx. These measurements are not always available from the EPA PAMS network. Cardelino and Chameides (2000) found that measured NO from the PAMS network was not precise enough to provide information about NOx-VOC sensitivity. Kleinman et al. (2000, 2001, 2002) used research-grade measurements.
These methods might also lead to erroneous results if measurement sites are affected by emissions in the immediate vicinity of the site. In addition, mixing ratios of reactive VOC such as isoprene may vary greatly with height, even within a daytime convective mixed layer (Andronache et al., 1994, Guenther et al., 1996a and b). Surface measurements may therefore not reflect the full range photochemical conditions that produce ozone.
[Top of section].
Parrish et al. (1998, 2000) described an analysis of measured correlations between ambient NOx and individual VOC species, which could be used for two purposes: (i) infer emission rates and ratios between emissions of individual VOC and NOx; and (ii) provide quality assurance tests for the accuracy and applicability of measured NOx and VOC. This work drew on earlier findings by Goldan et al. (1995, 1997) and Buhr et al. (1992, 1995).
Species correlations as a basis for estimating emissions have been investigated in Boulder, CO (Parrish et al., 1991, Goldan et al., 1995, 1997), Nashville, TN (Goldan et al., 2001), Los Angeles (Lurmann and Main, 1992), and at rural sites in the eastern U.S. (Buhr et al., 1992, 1995).
Parrish et al. (1998) and Goldan et al. (1995, 1997) reported that correlations between individual VOC (or among individual VOC and NOx or NOy) with similar atmospheric lifetimes should show a slope that is equal to the ratio of emission rates among the species (Figure 4.1). Correlations among individual VOC with different lifetimes were more complicated.
Parrish et al. (1998, 2000) also recommended a series of tests for internal consistency among sets of measured VOC. These were based on the expected correlation between species with common origin and similar atmospheric lifetimes. They also were based on the expectation that ratios among individual VOC changed from urban centers to downwind locations in a way that reflected the relative rates of photochemical loss.
Figure 4.2 shows ratios of individual VOC relative to a reference species from measurements at sites in the U.S., in comparison with ratios among individual VOC from emission inventories. The measured ratios are often lower than the emissions ratios because photochemical removal of species tends to lower the ratio. (The reference species is the least reactive). Measurements that match very closely to the emissions ratios, even for reactive species following photochemical aging, suggest the influence of emission sources in the immediate vicinity of the measurement site (Parrish et al., 2000).
For more information on VOC internal consistency tests: Go to
Parrish et al., 1998
[available here as PDF]
and
Parrish et al., 2000
[available here as PDF].
These methods are also related to techniques of receptor modeling (Henry, 1994, Kim and Henry, 2000, Watson et al. 2001).
None of these methods are applicable for isoprene, because ambient isoprene is not expected to correlate with anthropogenic species.
.jpg)
Figure 4.1. Measured correlations between Benzene and NOy during winter at an urban site (Boulder, CO) and at a rural site (Idaho Hill, CO). From Trainer et al. (2000) based on measurements reported by Goldan et al. (1995, 1997).
.jpg)
(a)
.jpg)
(b)
Figure 4.2. Log plots of ratios of (a) individual aromatics to benzene, and (b) individual alkenes to ethene, arranged in order of increasing reactivity or the aromatic or alkene. Each symbol and error bar gives the
geometric mean and standard deviation of the labeled ratio and colored data set. The color key is on the right. PAMS and research data sets are represented by circles and triangles respectively.
The symbol is closed only if more than 50% of both hydrocarbons in the ratio are above the detection limit. The solid line gives urban median ratios. From Parrish et al. (2000).
Update: The five-cities study by Kleinman et al. (2005) provided in-depth data on VOC and NOx in different urban areas. Kleinman et al. calculated instantaneous rates of photochemical production of ozone based on measured NO2, hydrocarbons and sunlight from measurements in each urban center. Results (Figures 4a1) show production rates increasing with NO2 up to a maximum value and then decreasing, comparable to the familiar isopleth plots. The transition from NOx-sensitive to VOC-sensitive conditions occurs when the ratio of reactivity-weighted VOC (in s-1) and NOx (in ppb) is 1. This also corresponds with Tonnessen's formulation, given above.
Results also show that the transition from NOx to VOC sensitive conditions correspond closely with the theoretical Ln/Q formulation, that represents the ratio between removal of NOx to the source of Hx radicals, that in theory is the cause of the transitiion.

(b)
Figure 4a.1.
Ozone production rates for five cities displayed as color-coded symbols as a function of VOCR and NOx concentration: (a) Nashville, (b) NYC, (c) Phoenix, (d) Philadelphia, and (e) Houston. Data are ordered from low values of P(O3) to high values, so high-P(O3) samples in some cases are hiding low-P(O3) samples underneath. Isolines of constant VOCR/NOx are shown by diagonal lines. From Kleinman et al. (2005)
[Top of section].
The following steps are proposed as a basis for evaluating ozone formation in an urban area based on a data set with measured VOC and either NOx or NOy. The evaluation should include four major components:
1. Evaluation of measured VOC for consistency, based Parrish et al. (1998, 2000). Recommended method: Plot the ratio of individual species relative to a reference hydrocarbon for each of the major reaction classes (alkanes, alkenes, aromatics), on comparison with standard ratios reported by Parrish et al., or with ratios from emission inventories for the specific metropolitan area.
2. Evaluation of measured VOC and NOx for comparison with emission inventories. Recommended method: Plot correlations of each VOC surrogate or reaction class versus a reference (e.g. alkanes) from the data set and from chemistry/transport models. Similarly, plot NOx or NOy versus a reference hydrocarbon class, and compare with the equivalent plot from chemistry/transport models.
3. Evaluation of isoprene. Recommended method: Plot the measured diurnal profile of isoprene in comparison with chemistry/transport model results. WARNING: Model-measurement discrepancies for isoprene (especially during the early morning or late afternoon) may be due to the rate of daytime vertical mixing in the chemistry/transport model. Isoprene in the model should show a vertical profile during midday that is comparable to previously measured vertical profiles (see Figure 4.3).
4. Evaluation for NOx-VOC sensitivity. Recommended method: Plot reactivity-weighted VOC versus NOx (or NOy)for all measurement sites in the metropolitan area, in comparison with results from photochemical models for the same locations. It may be useful to do this separately for measurements during the morning (which relate most closely to emission rates for anthropogenic species) and during midday and afternoon (which relate to photochemical formation).
This representation forms a surrogate evaluation for NOx-VOC sensitivity because the sensitivity of ozone production rates is related to NOx-VOC sensitivity. A consistent bias towards underprediction or overprediction would suggest that the model application may be biased in its NOx-VOC predictions.
Plots of rVOC versus NOx or NOy are advantageous because they can easily be used to evaluate chemistry/transport models. A consistent bias towards underprediction or overprediction would suggest that the model application may be biased in its NOx-VOC predictions. (Criteria are needed to evaluate model performance.)
Methods recommended by Kleinman et al. or Cardelino et al., discussed above, are equally valid alternatives for evaluating NOx-VOC sensitivity based on measured NOx and VOC.
Modifications based on measurement results: The evaluation of emission inventories may suggest a systematic underestimate or overestimate of one class of VOC, or of NOx. This might be used to generate an alternative model scenario with adjusted emission rates that would show better agreement with measurements. Mendoza-Dominguez and Russell (2000, 2001) have formalized this using inverse modeling.
If either NOy or HNO3 is available, the modified model scenario can be evaluated using indicator correlations (see Section 2). Results that show both improved agreement with both primary VOC and NOx and also better agreement with the secondary indicator correlations (O3 vs. NOy, O3 vs HNO3, etc.) would suggest that the alternative scenario is more accurate.
.jpg)
Figure 4.3. Isoprene versus altitude in meters. Isoprene is shown in units relative to surface concentrations. The solid dots represent the median profile from measurements at midday at Rose, AL (Andronache et al., 1994). The asterisks are measurements at Pellston, MI (Sillman et al., 2002). The solid and dashed lines represent model values.
[Return to Table of Contents]
Andronache, C., W. L. Chameides, M. O. Rodgers, J. E. Martinez, P. Zimmerman, and J. Greenberg. Vertical distribution of isoprene in the lower boundary layer of the rural and urban southern United States. J. Geophys. Res., 99,16989-17000, 1994.
Blanchard, C. L., and T. Stoeckenius, Ozone response to precursor controls: comparison of data analysis methods with the predictions of photochemical air quality simulation models. Atmos. Environ., 35, 1203-1216, 2001.
Blanchard, C. L., Ozone process insights from field experiments- Part III: Extent of reaction and ozone formation. Atmos. Environ. 2000, 34: 2035-2043.
Blanchard, C. L., F. W. Lurmann, P. M. Roth, H. E. Jeffries, and M. Korc, The use of ambient data to corroborate analyses of ozone control strategies, Atmos. Environ., 33, 369-381, 1999.
Buhr, M., D Parrish, J. Elliot, J. Holloway, J. Carpenter, P. Goldan, W. Kuster, M. Trainer, S. Montzka, S. McKeen, and F. C. Fehsenfeld, Evaluation of ozone precursor source types using principal component analysis of ambient air measurements in rural Alabama. J. Geophys. Res., 100, 22853-22860, 1995.
Buhr, M. P., M. Trainer, D. D. Parrish, R. E. Sievers, and F. C. Fehsenfeld, Assessment of pollutant emission inventories by principal component analysis of ambient air measurements, Geophys. Res. Letters, 19, 1009-1012, 1992.
Cardelino, C. and W. L. Chameides. An observation-based model for analyzing ozone-precursor relationships in the urban atmosphere. J. Air Waste Manage. Assoc., 45, 161-180, 1995.
Cardelino, C. A. and W. L. Chameides, The application of data from photochemical assessment monitoring stations to the observation-based model, Atmos. Environ., 34, 2325-2332, 2000.
Chameides, W. L., F. Fehsenfeld, M. O. Rodgers, C. Cardellino, J. Martinez, D. Parrish, W. Lonneman, D. R. Lawson, R. A. Rasmussen, P. Zimmerman, J. Greenberg, P. Middleton, and T. Wang, Ozone precursor relationships in the ambient atmosphere. J. Geophys. Res., 97, 6037-6056, 1992.
Chock, D. P., T. Y. Chang, S. L. Winkler, and B. I. Nance, The impact of an 8 h ozone air quality standard on ROG and NOx controls in Southern California. Atmos. Environ., 33, 2471-2486, 1999.
Duncan, B. N., Y. Yoshida, J. R. Olson, S. Sillman, R. V. Martin, L. Lamsal, Y. Hu, K. E. Pickering, C. Retscher, D. J. Allen, and J. H. Crawford, Application of OMI Observations to a Space-Based Indicator of NOx and VOC Controls on Surface Ozone Formation, Atmos. Environ., 44, 2213-2223, doi:10.1016/j.atmosenv.2010.03.010, 2010.
Gao, D., W. R. Stockwell and J. B. Milford, Global uncertainty analysis of a regional-scale gas phase chemical mechanism. J. Geophys. Res., 101 9071-9078, 1996.
Gillani, N. V. and J. E. Pleim. Sub-grid-scale features of anthropogenic emissions of NOx and VOC in the context of regional Eulerian models. Atmos. Environ., 30, 2043-2059, 1996.
Goldan, P. D., M. Trainer, W. C. Kuster, D. D. Parrish, J. Carpenter, J. M. Roberts, J. E. Yee, and F. C. Fehsenfeld. Measurements of hydrocarbons, oxygenated hydrocarbons, carbon monoxide and nitrogen oxides in an urban basin in Colorado: implications for emissions inventories. J. Geophys. Res., 100, 22771-22785, 1995.
Goldan, P. D., W. C. Kuster, and F. C. Fehsenfeld, Non-methane hydrocarbon measuremetns during the tropospheric OH photochemistry experiment, J. Geophys. Res., 102, 6315-6324, 1997.
Goldan, P. D., D. D. Parrish, W. C. Kuster, M. Trainer, S. A. McKeen, J. Holloway, B. T. Jobson, D. T. Sueper, and F. C. Fehsenfeld, Airborne measurements of isoprene, CO and anthropogenic hydrocarbons and their implications, J. Geophys. Res., in press, 2001.
Guenther, A., P. Zimmerman, L. Klinger, J. Greenberg, C. Ennis, K. Davis, W. Pollock, H. Westberg, G. Allwine, and C. Geron, Estimates of regional natural volatile organic compound fluxes from enclosure and ambient measurements. J. Geophys. Res., 101, 1345-1359, 1996a.
Guenther, A., W. Baugh, K. Davis, G. Hampton, P. Harley, L. Klinger, L. Vierling, P. Zimmerman, E. Allwine, S. Dilts, B. Lamb, H. Westberg, D. Baldocchi, C. Geron, and T. Pierce, Isoprene fluxes measured by enclosure, relaxed eddy accumulation, surface layer gradient, mixed layer gradient, and mixed layer mass balance techniques. J. Geophys. Res., 101, 18555-18567, 1996b.
Hammer, M.-U., B. Vogel, and H. Vogel, Findings on H2O2/HNO3 as an Indicator of Ozone Sensitivity in Baden-Wuerttemberg, Berlin-Brandenburg, and the Po Valley Based on Numerical Simulations, J. Geophys. Res., in press, 2001.
Henry, R. C., C. W. Lewis, P. K. Hopke, and H. W. Williamson, Review of receptor modeling fundamentals, Atmos. Environ., 18, 1507-1515, 1984.
Hirsch, A. I., J. W. Munger, D. J. Jacob, L. W. Horowitz, and A. H. Goldstein, Seasonal variation of the ozone production efficiency per unit NOx at Harvard Forest, Massachusetts. J. Geophys. Res., 101, 12659-12666, 1996.
Jacob, D. J., B. G. Heikes, R. R. Dickerson, R. S. Artz and W. C. Keene. Evidence for a seasonal transition from NOx- to hydrocarbon-limited ozone production at Shenandoah National Park, Virginia. J. Geophys. Res., 100, 9315-9324, 1995.
Jaegle, L., D.J. Jacob, W.H. Brune, D. Tan, I. Faloona, A.J. Weinheimer, B.A. Ridley, T.L. Campos, and G.W. Sachse, Sources of HOx and production of ozone in the upper troposphere over the United States, Geophys. Res. Lett., 25, 1705-1708, 1998.
Kim, B. M. and R. C. Henry, Application of SAFER model to tbe Los Angeles PM10 data, Atmos. Environ., 34, 1747-1759, 2000.
Kirchner, F., F. Jeaneret, A. Clappier, B. Kruger, H. van den Bergh, and B. Calpini, Total VOC reactivity in the planetary boundary layer 2. A new indicator for determining the sensitivity of the ozone production to VOC and NOx, J. Geophys. Res., 106, 3095-3110, 2001.
Kleinman, L. I., Daum, P. H., Lee, Y-N., Nunnermacker, L. J., Springston, S. R., Newman, L., Weinstein-Lloyd, J., and Rudolph, J.: A comparative study of ozone production in five U.S. metropolitan areas, J. Geophys. Res., 110, D02301, doi:10.1029/2004JD005096, 2005.
Kleinman, L. H., P. H. Daum, Y-N. Lee, L. J. Nunnermacker, S. R. Springston, J. Weinstein-Lloyd, and Jochen Rudolph, Sensitivity of ozone production rate to ozone precursors, Geophys. Res. Lett.,28, 2903-2906, 2001, 2001.
Kleinman, L. I., P. H. Daum,Y-N. Lee, L. J. Nunnermacker, S. R. Springston, J. Weinstein-Lloyd, P. Hyde, P. Doskey, J. Rudolf, J. Fast and C. Berkowitz, Photochemical age determinations in the Phoenix metropolitan area. J. Geophys. Res., 10.1029/2002JD002621, 2003.
Kleinman, L. I., P. H. Daum, D. G. Imre, J. H. Lee, Y-N. Lee, L. J. Nunnermacker, S. R. Springston, J. Weinstein-Lloyd, and L. Newman, Ozone production in the New York City urban plume, J.Geophys. Res., 105, 14495-14511, 2000.
Kleinman, L. I., Ozone procuess insights from field experiments - part II; observation-based analysis for ozone production. Atmos. Environ., 34, 2023-2034, 2000.
Kleinman, L. I., P. H. Daum, J. H. Lee, Y-N. Lee, L. J. Nunnermacker, S. R. Springston, L. Newman, J. Weinstein-Lloyd and S. Sillman. Dependence of ozone production on NO and hydrocarbons in the troposphere. Geophys. Res. Lett., 24, 2299-2302, 1997.
Kleinman, L. I., Low and high-NOx tropospheric photochemistry. J. Geophys. Res., 99, 16831-16838, 1994.
Lin, X., M. Trainer, and S. C. Liu, On the nonlinearity of tropospheric ozone, J. Geophys Res., 93, 15879-15888, 1988.
Liu, S. C., M. Trainer, F. C. Fehsenfeld, D. D. Parrish, E. J. Williams, D. W. Fahey, G. Hubler, ad P. C. Murphy, Ozone production in the rural troposphere and the implications for regional and global ozone distributions. J. Geophys. Res., 92, 4191-4207, 1987.
Lu, C-H. and J. S. Chang. On the indicator-based approach to assess ozone sensitivities and emissions features. J. Geophys. Res., 103, 3453-3462, 1998.
Luke, W. T., T. B. Wtson, K. J. Olszyna, R. Laureen Gunter, R. T. McMillen, D. L. Wellman, and S. W. Wilkison, Comparison of airborne and surface trace gas measurements during the Southern Oxidant Study, J. Geophys. Res., 103, 22317-22337, 1998.
Martilli, A., A. Neftel, G. Favaro, F. Kirchner, S. Sillman, and A. Clappier, Simulation of the ozone formation in the northern part of the Po Valley with the TVM-CTM. J. Geophys. Res., in press, 2001.
McClenny, W. A. ed, Recommended Methods for Ambient Air Monitoring of NO, NO2, NOy, and Individual NOz Species, EPA/600/R-01/005, September, 2000.
Mendoza-Dominguez, A., J. W. Boylan, Y-J. Yang and A. G. Russell, Efficient sensitivity analysis of an air quality model for primary and secondary aerosol source impact quantification, submitted to J. Geophys. Res., 2002.
Mendoza-Dominguez, A., and A. G. Russell, Estimation of emission adjustments from the application of four-dimensional data assimilation to photochemical air quality modeling, Atmospheric Environment, 35, 2879-2894, 2001.
Mendoza-Dominguez and Russell, Iterative Inverse Modeling and Direct Sensitivity Analysis of a Photochemical Air Quality Model, Environ. Sci. Technol., 3, 4974-4981, 2000.
Milford, J., D. Gao, S. Sillman, P. Blossey, and A. G. Russell. Total reactive nitrogen (NOy) as an indicator for the sensitivity of ozone to NOx and hydrocarbons. J. Geophys. Res. , 99, 3533-3542, 1994.
Milford, J., A. G. Russell, and G. J. McRae, A new approach to photochemical pollution control: implications of spatial patterns in pollutant responses to reductions in nitrogen oxides and reactive organic gas emissions. Environ. Sci. Tech. 23, 1290-1301, 1989.
NARSTO. An Assessment of Tropospheric Ozone Pollution: A North American Perspective. The NARSTO Synthesis Team, July, 2000.
Parrish, D. D., et al., Internal consistency tests for evaluation of measurements of anthropogenic hydrocarbons in the troposphere, J. Geophys. Res.,, 103, 22,339-22,359, 1998.
Parrish, D.D., and F.C. Fehsenfeld, Methods for gas-phase measurements of ozone, ozone precursors and aerosol precursors, Atmos, Environ., 34, 1921-1957, 2000.
Pierce, T., C. Geron, L. Bender, R. Dennis, G. Tonnesen, and A. Guenther, Influence of increased isopreneemissions on regional ozone modeling, J. Geophys. Res.,103, 25611-25630, 1998.
Reynolds, S., H. Michaels, P. Roth, T. W. Tesche, D. McNally, L. Gardner, and G. Yarwood, Alternative base cases in photochemical modeling: their construction, role, and value. Atmos. Environ., 30, 12, 1977-1988, 1996.
Sillman, S. The use of NOy, H2O2 and HNO3 as indicators for O3-NOx-ROG sensitivity in urban locations. J. Geophys. Res., 100, 14175-14188, 1995.
Sillman, S., K. Al-Wali, F. J. Marsik, P. Nowatski, P. J. Samson, M. O. Rodgers, L. J. Garland, J. E. Martinez, C. Stoneking, R. E. Imhoff, J-H. Lee, J. B. Weinstein-Lloyd, L. Newman and V. Aneja. Photochemistry of ozone formation in Atlanta, GA: models and measurements. Atmos. Environ., 29, 3055-3066, 1995.
Sillman, S., D. He, C. Cardelino and R. E. Imhoff. The use of photochemical indicators to evaluate ozone-NOx-hydrocarbon sensitivity: Case studies from Atlanta, New York and Los Angeles. J. Air Waste Manage. Assoc., 47, 642-652, September, 1997.
Sillman, S., D. He, M. Pippin, P. Daum, L. Kleinman, J. H. Lee and J. Weinstein-Lloyd. Model correlations for ozone, reactive nitrogen and peroxides for Nashville in comparison with measurements: implications for VOC-NOx sensitivity. J. Geophys. Res. 103, 22629-22644, 1998.
Sillman, S., M. T. Odman, and A. G. Russell, Comment on "On the indicator-based approach to assess ozone sensitivities and emissions features" by C-H. Lu and J. S. Chang, J. Geophys. Res., 106 , D18 , 20,941, 2001.
Sillman, S., Comment on 'The Impact of An 8-hour Ozone Air Quality Standard on VOC and NOx Controls in Southern California' by Chock, et al., Atmos. Environ. 35, 3370-3371, 2001.
Sillman, S., and D. He, Some theoretical results concerning O3-NOx-VOC chemistry and NOx-VOC indicators, J. Geophys. Res., 107, D22, 4659, doi:10.1029/2001JD001123, 2002.
Sillman, S., Carroll, M. A., Thornberry, T., Lamb, B. K., Westberg, H., Brune, W. H., Faloona, I., Tan, D., Hurst, J. M., Shepson, P. B., Sumner, A., Hastie, D. R., Mihele, C. M., Apel, E. C., Riemer, D. D., and Zika, R. G. Loss of isoprene and sources of nighttime OH radicals at a rural site in the U.S.: Results from photochemical models. J. Geophys. Res. , 107 (D5), 10.1029/2001JD000449, 2002.
Sillman, S., R. Vautard, L. Menut and D. Kley, O3-NOx-VOC sensitivity and NOx-VOC indicators in Paris: results from models and ESQUIF measurements, J. Geophys. Res., 108, 8563 doi:10.1029/2002JD001561, 2003.
Sillman, S. and West, J. J.: Reactive nitrogen in Mexico City and its relation to ozone-precursor sensitivity: results from photochemical models, Atmos. Chem. Phys., 9, 3477-3489, 2009 (available at http://www.atmos-chem-phys.net/9/3477/2009/acp-9-3477-2009.html).
Sosa, G., J. West, F. San Martini, L. T. Molina and M. J. Molina, "Air Quality Modeling and Data Analysis for Ozone and Particulates in Mexico City." MIT Integrated Program on Urban, Regional and Global Air Pollution Report No. 15, 76 pages, October 2000, available from http://eaps.mit.edu/megacities/index.html.
Thielman, A., A. S. H. Prevot, and J. Staelhelin, Sensitivity of ozone production derived from field measurements in the Italian Po Basin, J. Geophys. Res., in press, 2001b.
Tonnesen, G. S. and R. L. Dennis, Analysis of radical propagation efficiency to assess ozone sensitivity to hydrocarbons and NOx. Part 1: Local indicators of odd oxygen production sensitivity, J. Geophys. Res., 105, 9213-9225, 2000a.
Tonnesen, G. S., and R. L. Dennis, Analysis of radical propagation efficiency to assess ozone sensitivity to hydrocarbons and NOx. Part 2:Long-lived species as indicators of ozone concentration sensitivity, J. Geophys. Res., 105, 9227-9241, 2000b.
Trainer, M., D. d. Parrish, P. d. Golday, J. Roberts, and F. C. Fehsenfeld, Review of observation-based analysis of the regional factors influencing ozone concentrations, Atmos. Environ., 34, 2045-2061, 2000.
Trainer, M., D. D. Parrish, M. P. Buhr, R. B. Norton, F. C. Fehsenfeld, K. G. Anlauf, J. W. Bottenheim, Y.Z. Tang, H.A. Wiebe, J.M. Roberts, R.L. Tanner, L. Newman, V.C. Bowersox, J.M. Maugher, K.J. Olszyna, M.O. Rodgers, T. Wang, H. Berresheim, and K. Demerjian. Correlation of ozone with NOy in photochemically aged air. J. Geophys. Res., 98, 2917-2926, 1993.
Vogel, B., N., Riemer, H. Vogel, F. Fiedler, Findings on NOy as an indicator for ozone sensitivity based on different numerical simulations, J. Geophys. Res., 3605-3620, 1999.
Watson, J. G., J. C. Chow, and E. M. Fujita, Review of volatile organic compound source apportionment by chemical mass balance, Atmos. Environ., 35, 1567-1584, 2001.
[Top of page]

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
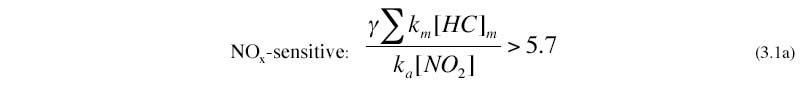
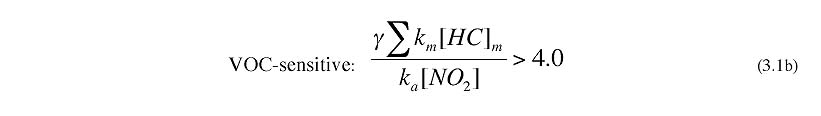
.jpg)
.jpg)
.jpg)
.jpg)