 |
|
| Mole Balance: | 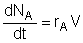

|
| Rate Law: | 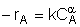 |
| Stoichiometry: | 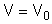 |
| Combine: | 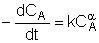 |
Consider the following reaction that occurs in a constant volume batch reactor: (We will withdraw samples and record the concentration of A as a function of time.
Mole Balance: 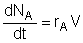



Rate Law: Stoichiometry: Combine:
Differential Method
Taking the natural log of The reaction order can be found from a ln-ln plot of:
Three Ways to Determine -dCA/dt
Methods for finding the slope of log-log and semi-log graph papers may be found here.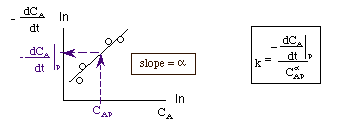
2. Polynomial (using Polymath)
CA = ao + a1t + a2t2 + a3t3 +a4t4
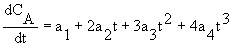
3. Numerical
Finite Difference
Non-Linear Least-Squares Analysis
Generally want
to be a minimum.
For concentration-time data,
Integral Method
We find the values of alpha and k which minimize S2
Finally we should also use the formula to plot reaction rate data in terms of concentration vs. time for 0, 1st, and 2nd order reactions.
Table CD5-1
These types of plots are usually used to determine the values k for runs at various temperatures and then used to determine the activation energy.
Zero Order First Order Second Order
 "What is Wrong with This Solution?" - Chapter 5
"What is Wrong with This Solution?" - Chapter 5
* All chapter references are for the 3rd Edition of the text Elements of Chemical Reaction Engineering.