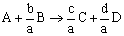
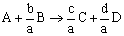
Using stoichiometry, we set up all of our equations with the amount of reactant A as our basis.
Batch System Stoichiometric Table :
Species Symbol Initial Change Remaining A A B B C C D D Inert I
________-------
____________
Where:
and
Concentration -- Batch System:
Constant Volume Batch:
[a liquid phase reaction, or a gas phase reaction in a rigid (e.g., steel) reactor] etc.
Flow System Stoichiometric Table :
Species Symbol Reactor Feed Change Reactor Effluent A A B B C C D D Inert I
________-------
____________
Where:
and 

Concentration -- Flow System:
Liquid Phase Flow System:
etc.
If the rate of reaction werethen we would have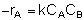 This gives us -rA = f(X). Consequently, we can use the methods discussed in Chapter 2 to size a large number of reactors, either alone or in series.
This gives us -rA = f(X). Consequently, we can use the methods discussed in Chapter 2 to size a large number of reactors, either alone or in series.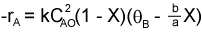
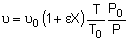




etc.
Again, these equations give us information about -rA = f(X), which we can use to size reactors.Matching CA versus X curves