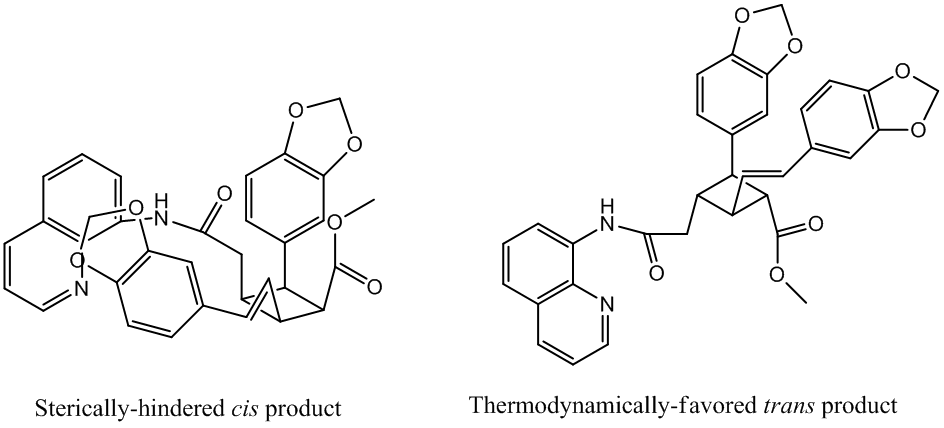
(3) the sequential functionalization product 16 in hand, the relative stereochemistry needed to be altered to the all-trans configuration found in the natural product. This was anticipated to be a facile process given the strained nature of the all-cis stereochemistry and the thermodynamically downhill path to the desired all-trans product” (7509).
(4) “These [pipercyclobutanamide] heterodimers represent a greater synthetic challenge than the piperarborenines (1,3) because of the presence of four different substituents on the cyclobutane ring. Both of these natural products contain an unusual cis unsaturated amide…” (7507).
Source paper: Gutekunst, W. R.; Gianatassio, R.; Baran, P. S. Angew. Chem. Int. Ed. 2012, 51, 7507-7510.
University of Michigan Chem 215/216 HH Winter 2014. Nicholas Carducci's Structured Study Group. HTML Project of Callie Chappell, James Lawniczak, Aiman Faruqi, and Ryan Gentil