Chapter 5: Isothermal Reactor Design: Conversion
Batch Reactor With a Reversible Reaction
The following reaction follows an elementary rate law
![]()
Initially 77% N2, 15% O2, 8% inerts are fed to a batch reactor where
80% of the equilibrium conversion (Xe = 0.02) is reached in 151 ![]() s.
What is the specific reaction rate constant k1?
s.
What is the specific reaction rate constant k1?
Additional Information
![]()
Solution
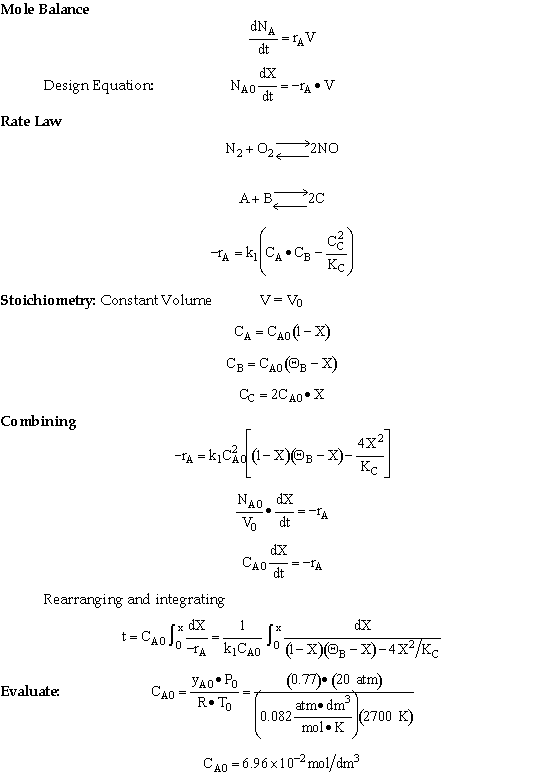
For 80% of equilibrium conversion X = 0.8 Xe = 0.016
![]()
![]()
![]()
Use Simpson's three point formula to integrate with ΔX = 0.016/2 = 0.008
![]()
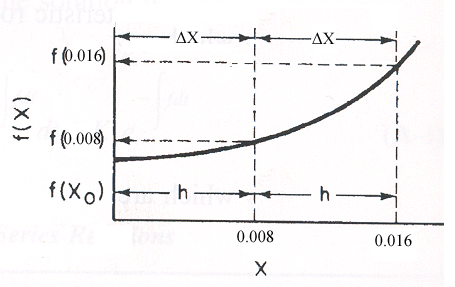
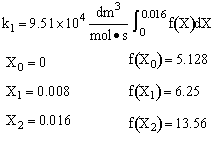
Simpson's rule: (ΔX = h = 0.008)
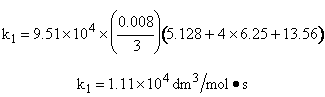
Back to Chapter 5