Chapter 8: Multiple Reactions
Additional Homework Problems
CDP6-BB
The production of maleic anhydride by the air oxidation of benzene was recently studied using a vanadium pentoxide catalyst. [Chem. Eng. Sci., 43, 1051 (1988).]
The reactions that occur are:
| Reaction 1: | C6H6 + (9/2)O2 |
| Reaction 2: | C4H2O3 + 3O2 |
| Reaction 3: | C6H6 + (15/2)O2 |
Because these reactions were carried out in excess air, volume change with reaction can be neglected, and the reactions can be written symbolically as this reaction sequence, where Reaction 1 is second-order and all others are first-order:
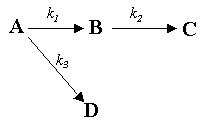
A = Benzene, B = Maleic anhydride, C = Products (H2O, CO2), and D = Products (CO2, H2O)
The specific reaction rates at the operating temperature are:
k1 = 0.2 (dm6/mol/kg cat/s)
k2 = 4*10-4 (dm6/kg cag/s)
k3 = 6*10-4 (dm6/kg cat/s)
These reactions are carried out isothermally in both a CSTR and a PBR. Benzene enters the reactor at a concentration of 0.01 mol/dm3, and the total volumetric flow rate is 2.5 dm3/s.
| (a) | For a catalyst weight of 5000 kg, determine the exit concentrations from a "fluidized" CSTR. |
| (b) | What is the selectivity of B to C and of B to D in the CSTR? |
| (c) | Plot the concentrations of all species as a function of PBR catalyst weight in a PBR. Determine the catalyst weight for which the concentration of B is a maximum. |
| (d) | What is the effect of the entering concentration of benzene on your results. How would your product distribution change if CAo = 0.5 or 0.001 mol/dm3? |