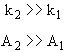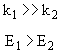Chapter 8: Multiple Reactions
What Type of Reaction is Taking Place?
Three species were found in a CSTR. The following concentration data were
obtained as a function of temperature. The initial concentration of the single
reactant, A, was the same at all temperatures. Both B and C are products.
CA0 = 2moles/dm3
|
Run
|
T (oC)
|
CA (mole/dm3)
|
CB (mole/dm3)
|
CC (mole/dm3)
|
|
1
|
30
|
1.7
|
0.01
|
0.29
|
|
2
|
50
|
1.4
|
0.03 |
0.57
|
|
3
|
70
|
1.0
|
0.1
|
0.90
|
|
4
|
100
|
0.5
|
1.25
|
1.25
|
|
5
|
120
|
0.1
|
1.80
|
0.1
|
|
6
|
130
|
0.01
|
1.98
|
0.01
|
Explain the above data, what type of reaction is taking place, independent,
complex, series or parallel ?
Hint 1: What are the trends?
Hint 2: What do the trends suggest?
Hint 1
Observations
At low temperatures
1) Little conversion of A
2) Little B formed
3) Mostly C formed (but not too much because of the low conversion - 15 to 30% - of A)
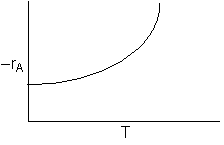
At high temperatures
1) Virtually complete conversion of A
2) Mostly B formed
Hint 2
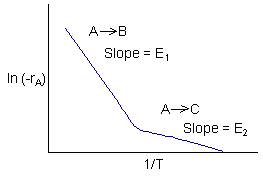
Data suggest 2 reactions
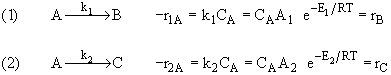
|
Solution
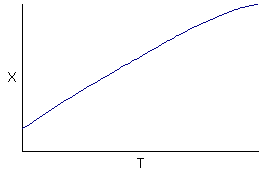
Observations
At low temperatures
1) Little conversion of A
2) Little B formed
3) Mostly C formed (but not too much because of the low conversion - 15 to 30% - of A)
At high temperatures
1) Virtually complete conversion of A
2) Mostly B formed
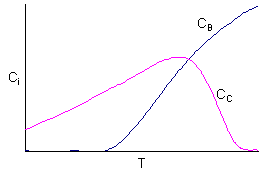
Data suggest 2 reactions
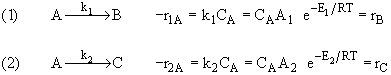
|
Reaction (1) is dominant at high temperatures
|
|
with
|
|
Reaction (2) is dominant at low temperatures
|
|
|
|
|
|
|
|