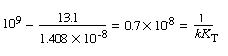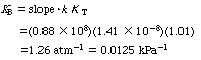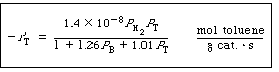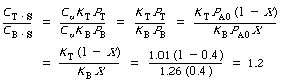Chapter 10: Catalysis and Catalytic Reactors
Learning Resources
Example CD10-2: Least Squares Analysis to Determine the Rate Law Parameters k, K T, and K B
(Example 6-2 in 2nd ed.)Use the data in Table 10-6 together with Equation (10-81) to evaluate the rate law parameters. Then calculate the catalyst weight necessary for these rate law parameters. |
|||
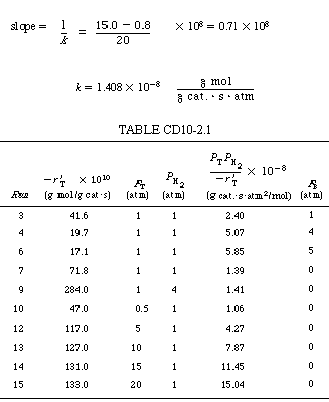
SolutionThe calculations are shown in Table CD10-2.1. A plot of |
|||
|
|
|||

Figure CD10-2.1 |
|||
Rearranging Equation (10-81) for PB = 0, we have |
|||
|
|
(CD10-2.1) |
||
| From Figure CD10-2.1, |
|||
|
|
|||
| Substituting these values into Equation (CD10-2.1), we find that |
|||
|
|
(CD10-2.2) |
||
The constant K B can be evaluated
from the slope of the plot of |
|||
|
|
|||

Figure CD10-2.2 |
|||
In addition to the graphical determination, we can use linear regression
to determine the rate law parameters. We will use Equations (5-31) through (5-33)
along with Table CD10-2.2 to determine k, K T , and |
|||
|
|
(10-81) |
||
and Equation (5-30), |
|||
|
|
(5-30) |
||
where Y= |
|||
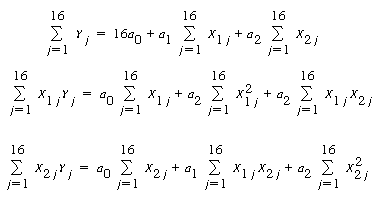
|
(CD10-2.3) |
||
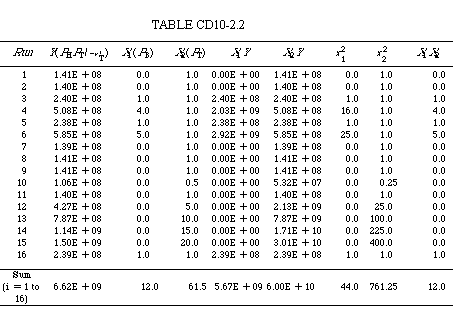
| If we were to use a software package such as POLYMATH, we would simply enter Y, X 1 X 2 , and for each run and the parameters a 0 , a 1 , and a 2 would be displayed in a few seconds. Alternatively, we can form Table CD10-2.2 and carry out the numerical operations ourselves to determine the rate law parameters. We can either form the table using a calculator or by using a spreadsheet such as Excel or Lotus 123. |
|||
|
|||
Equation (CD10-2.3) becomes |
|||
|
6.62 X 109 = 16a0 + 12a1 + 61.5a2 |
(CD10-2.6) |
||
Equation (CD10-2.4) becomes |
|||
|
5.67 X 109 = 12a0 = 44a = 12a21 |
(CD10-2.7) |
||
| Equation (CD10-2.5) becomes |
|||
|
6.0 X 1010 = 61.5a0 + 12a1 + 761.25a2 |
(CD10-2.8) |
||
| Solving these three equations simultaneously, we obtain a0 = 7.12 x 10 7 , a 1 = 9.0 x 10 7 , and a 2 = 7.16 x 10 7 . The corresponding rate law parameters are |
|||
| Using POLYMATH, we obtain |
|||
|
|
|||
|
with, of course, the parameter values being the same as those obtained
from Table CD10-2.2 and Equation (CD10-2.5).
is given by the equation |
|||
|
|
(CD10-2.9) |
||
whereP i is in atm. |
|||
After we have the adsorption constant K T and K B ,
we could calculate the ratio of sites. For example, the ratio of toluene sites to
benzene sites at 40% conversion is
|
|||
We see that at 40% conversion there are approximately 20% more sites occupied by toluene than by benzene. |
|||
Calculate the catalyst weight necessary for these parameters: |
|||
If we were to neglect pressure drop, we can solve for the catalyst weight necessary to achieve 65% conversion with the aid of Simpson's five-point formula. |
|||
|
|
|||
| Substituting for k, K T , K B ,P B , PH 2, and PT in Equation (E10-3.2) yields |
|||
|
|
(CD10-2.10) |
||
| We can now proceed to solve for the catalyst weight necessary to obtain this conversion by using Equation (CD10-2.10). |
|||
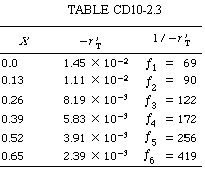
Hand calculations: The calculations for X versus (1/ |
|||
|
|
|||
Using numerical integration (Appendix A.5), after dividing the area under the curve into two parts: X = 0 to X = 0.52 with h 1 = 0.13 and X = 0.52 to X = 0.65 with h 2 = 0.13, we have |
|||
| Numerical |
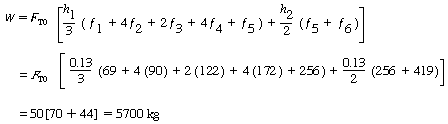 |
||
Since the reciprocal of the rate of reaction increases sharply as the conversion approaches unity, the numerical integration probably should have included a greater number of integrals than the six used in the calculation. |
|||