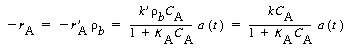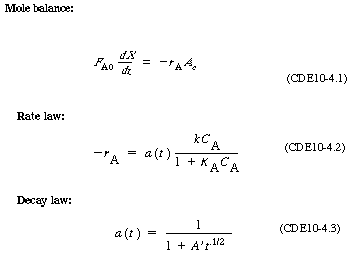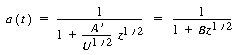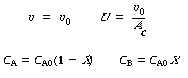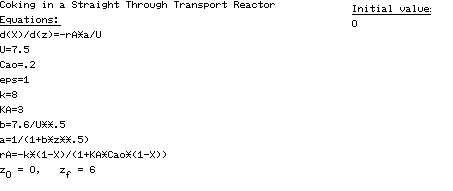Chapter 10: Catalysis and Catalytic Reactors
Learning Resources
Example CD10-3: Decay in a Straight-Through Reactor
The gas-phase cracking of a Salina light gas-oil reaction |
|||
|
A typical cost |
gas-oil (g) |
||
| is to be carried out in a straight-through transport reactor containing
a catalyst that decays as a result of coking. The reaction is carried out at 750°F. |
|||
|
|
|||
with K A = 3 m 3 /kmol and K
B = 0.01 m 3
/kmol. The maximum value of the term K B
C B is
small (0.002), so it can be neglected with respect to the other terms (e.g., 1).
Using this fact and the bulk density, |
|||
|
|
|||
| with k = 8 s -1 . At 750°F, the catalyst activity for light gas-oil over a synthetic catalyst for short contact times (i.e., less than 100 s) is |
|||
|
|
|||
| with A ´= 7.6 s -1/2 .
As a first approximation, neglect volume change with reaction, pressure drop, and
temperature variations. |
|||
Solution |
|||
|
|
|||
| For a catalyst particle traveling with a velocity of U, the
time that the catalyst particle has been in the reactor when it reaches a height |
|||
|
|
(CDE10-4.4) |
||
| and |
|||
|
|
(CDE10-4.5) |
||
| where |
|||
Stoichiometry. Gas-phase reaction with |
|||
|
|
|||
| Combining yields |
|||
|
|
(CDE10-4.6) |
||
| POLYMATH Solution. In using POLYMATH to obtain a solution, it is usually easiest to write the mole balance, rate law, and stoichiometry separately rather than combining them into a single equation. Therefore, starting with the mole balance |
|||
|
|
(CDE10-4.7) |
||
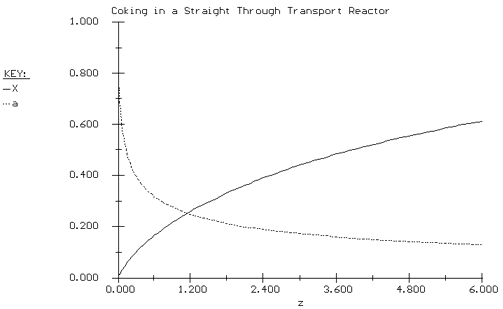
| we substitute for -r A and a using Equations (CDE10-4.2) and (CDE10-4.5). The POLYMATH program and solution are shown in Table CDE10-4.1 and Figure CDE10-4.1 respectively. |
|||
| Table CDE10-4.1 |
|||
|
|||
|