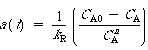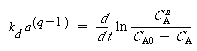Chapter 10: Catalysis and Catalytic Reactors
Professional Reference Shelf
R10.3-A Differential Method of Analysis to Determine the Decay Law
| The differential method of analysis can also be used to determine the order of decay for experiments carried out in either a CSTR or a plug-flow reactor. To illustrate this method, we consider a stirred contained solids reactor (SCSR), in which we measure the concentrations as a function of time. We assume the decay rate law to be of the form | |||
|
|
(CD10-16) | ||
| The design equation for the SCSR is the same as that for a CSTR. Consequently, for the n th-order irreversible reaction | |||
|
|
|||
| the activity at any time t is given by | |||
|
|
(CD10-17) | ||
| Taking the log and differentiating with respect to time, we get | |||
|
|
(CD10-18) | ||
| Combining Equations (10-136) and (10-137) yields | |||
|
|
(CD10-19) | ||
| Substituting Equation (10-141) for a and taking the ln of both sides gives us | |||
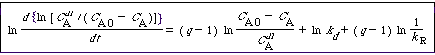
|
(CD10-20) |
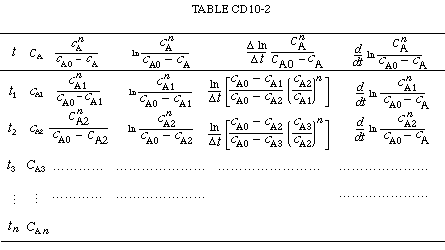
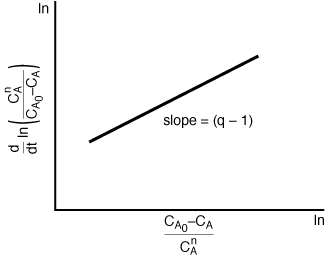
| We see that the reaction order can be found from a plot on log-log paper similar to that shown in Figure CD10-4. The data to construct this plot could be arranged as shown in Table CD10-2. By plotting the last column as a function of the reciprocal of the second column on log-log paper the decay order q can be obtained. | |||
|
|
|||
|