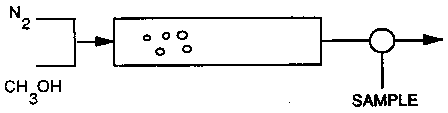
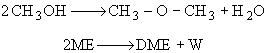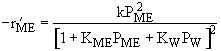Chapter 10: Catalysis and Catalytic Reactors
Dimethyl Ether
Self Test 1
|
|
|
|
|
|
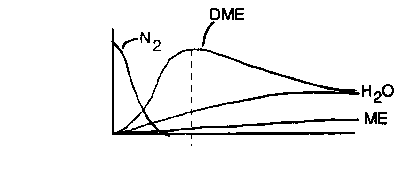
1) Why doesn't water initially exit the reactor the same as DME? Which of the following best describes the data? |
|
Self Test 2 - Dimethyl Either
|
|
|
|
|
|
|
2) Which of the following are true? The concentration of DME reaches a maximum because |
|
Solution
Self Test 3 Dimethyl
Either
| If the rate law is |
|
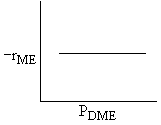
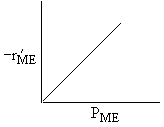
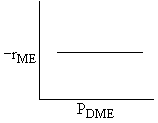
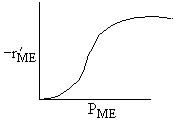
Which set of figures describes the rate the functionality of the partial pressure of this rate law.
| A | 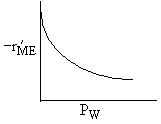
|

|
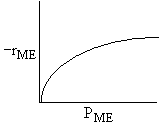
|
| B | 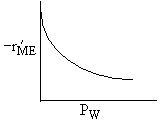
|

|
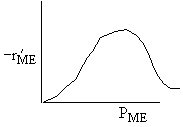
|
| C | 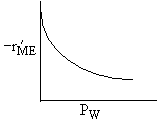
|
|
|
| D | 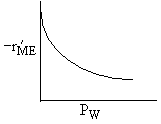
|
|
|
| No ME exits the reactor so it is either being consumed to form DME or being adsorbed on the surface or both. |
|
One mole of DME and one mole of water are formed for every two moles of ME consumed. DME exits the reactor while water does not, consequently it must be adsorbed on the surface. |
| We note when we reach steady state after a period of time, the exit concentration of DME and water are the same. Steady state has indeed been reached because the concentrations of W and DME are equal and the concentrations are no longer changed with time. |
Ans: E
|
F All of the above. |
| For every DME formed one water is formed. DME initially comes out the end of the reactor, while water does not. If DME were on the surface it would take up sites and would not exit for a while. The concentration of DME reaches a maximum because it initially has so many sites on which to react. After a while the water occupies a number of the sites so that there are fewer for ME to adsorb upon and react. |
| W : | 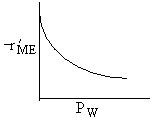
|
|
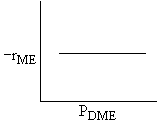
| DME: | 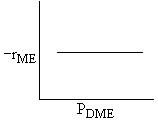
|
|
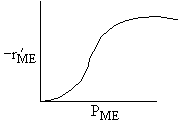
| ME : | 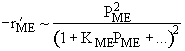
|
|
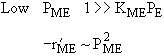
|
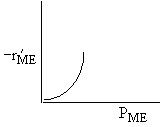
|
|
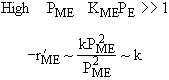
|
|
|
| Combining | ||
|
||
| Answer D |