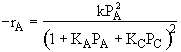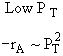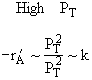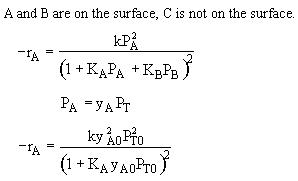Chapter 10: Catalysis and Catalytic Reactors
Example Exam Questions
Question 1

|
Figure 1: Data from a differential reactor. |
|
Which of the following best describe the data in the above figure?
|
|
|
Assume the reaction is irreversible |
|
|
What can you tell from the above figures? |
|
|
![]()
The following data were reported for the reaction
![]()
| A | B |
|

|
| C | D |

|

|
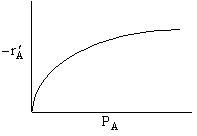
Which of the following mechanism is consistent with the above data
| (a) | (b) | (c) |
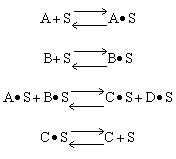
|

|
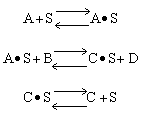
|
| (d) | (e) | |
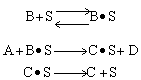
|

|
Question 4
![]()
The initial rate of reaction is shown below
| (a) | (b) | (c) |
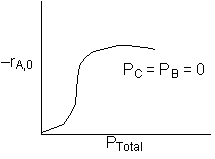
|
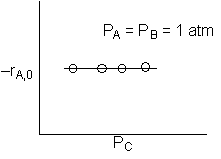
|
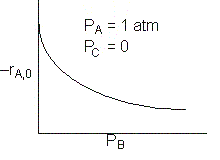
|
|
|
Question 5
![]()
The initial rate of reaction is shown below
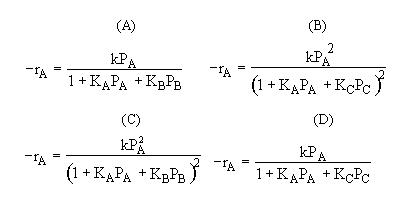
1. The rate law is
For the reaction![]() from Self Tests 12 and 13 we
found
from Self Tests 12 and 13 we
found
|
|
which mechanism is consistent with the rate law
|
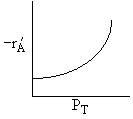
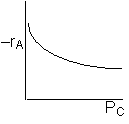
If the reaction is reversible the increase in PC will decrease the rate. If C is on the surface it will be in the denomination of the rate law and thus increasing PC will decrease the rate. Ans: C |
|
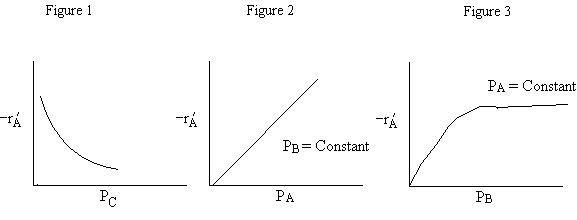
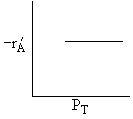
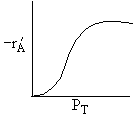
A is not on the surface or if it is KAPA << 1. B is on the surface and we see as we increase PB, the rate reaches a plateau. The curves from A, B, and C partial pressure suggest a rate law of the for |
|
Figure 1 |
|
Figure 2 |
|
Figure 3 |
|
Combining |
|
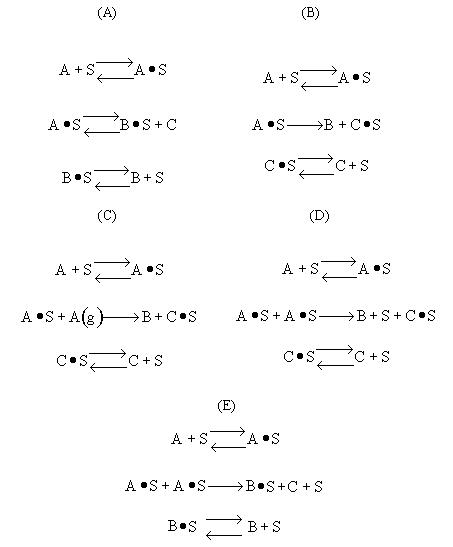
Possible Mechanism |
|
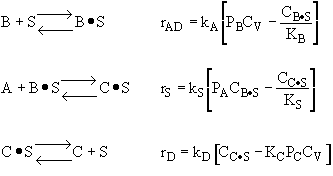
|
|
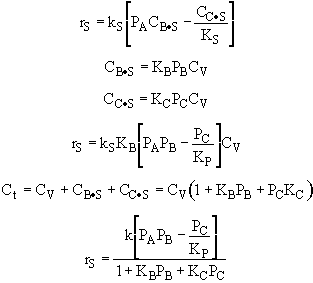
|
|
From the rate law we know species B and C are adsorbed on the surface. From the data you can’t tell whether or not C is adsorbed on the surface. |
|
Ans: E |
![]()
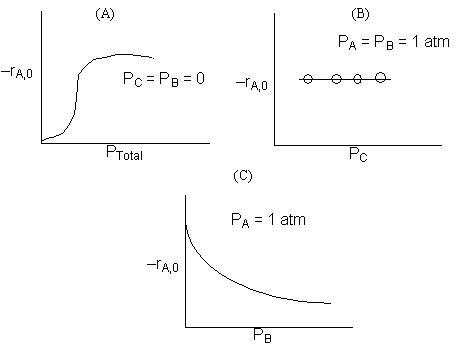
Recall |
|||||||||||||
Figure (a) suggests |
|||||||||||||
Figure (b) suggests |
|||||||||||||
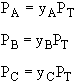
|
|||||||||||||
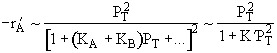
|
|||||||||||||
|
|||||||||||||
Figure (c) suggests |
|||||||||||||
|
|
|||||||||||||
Figure (d) suggests |
|||||||||||||
Combining all the above |
|||||||||||||
Therefore (b) is consistent |
|||||||||||||
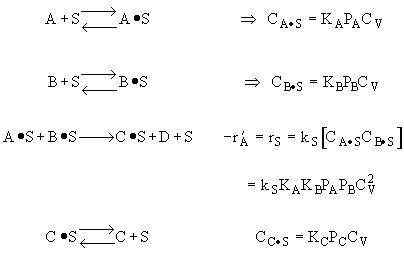
|
|||||||||||||
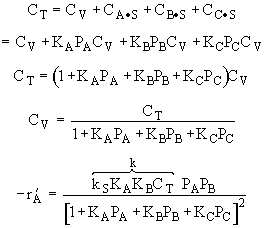
|
|||||||||||||
1. The reaction is irreversible because when PB = 1 atm, increasing the product C does not change the rate. (TRUE) |
2. Species B is on the surface because in 1 we showed that the reaction is irreversible, therefore the only way the rate could decrease were if were in the denominator of the rate law. If a species is in the denominator of the rate law it’s on the surface. (TRUE) |
3. Species C is not on the surface because increasing PC does not affect the rate.(FALSE) |
Ans: A |
|
|
|
Low PT |
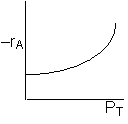
|
|
High PT |
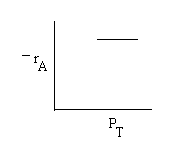
|
|
Combining |
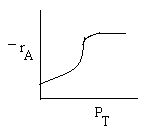
|
|

|
|
|
Ans: (c) |
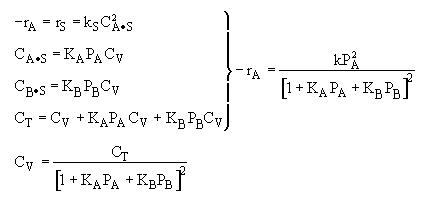
Ans: (e)