Chapter 13: Unsteady State Nonisothermal Reactor Design
Example DVD13-3: Falling Off The Steady State
Propylene glycol is produced in a CSTR in which precautions have been taken to prevent product loss. Propylene oxide (A) is fed to a 413-gal reactor at a rate of 300 lb mol/h at a temperature of 75°F. Water (B) is fed at a rate of 2000 lb mol/h and methanol at a rate of 20 lb mol/h. Determine what happens to a CSTR operating at 172°F and a conversion of 86.4% when there is a drop in feed temperature from 75°F to 68°F. |
||
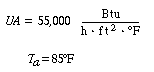 |
||
|
||
Solution |
||
We will first look at the steady-state conditions before the upset occurred. The mole balance conversion is: |
||
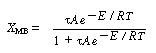 |
|
|
The energy balance conversion is: |
||
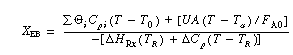 |
|
|
Neglecting |
||
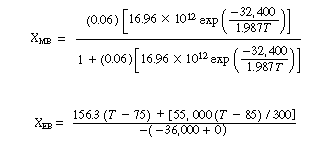 |
(CDE13-3.1) |
|
If we were to plot XEB and XB as a function of temperature, we would we see that there are three steady-state conditions for this set of parameter values (Table CDE13-3.1). Before the drop in feed temperature occurred we operated at the upper steady state (T = 172°F, X = 0.864). The ignition temperature of the feed is 78°F, and the extinction temperature is 61°F . |
||

|
||
We will now consider what happens when an upset occurs while
operating at the upper steady state. Say that the inlet temperature drops
from 75°F to 68°F. Figure CDE13-3.1 shows the corresponding XEB
and XMB curves after this drop.
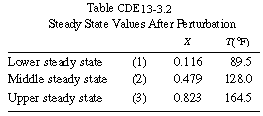
There are still three steady states (Table CDE13-3.2) for this new inlet
temperature, indicating that we have not dropped below the extinction temperature
of 61°F. 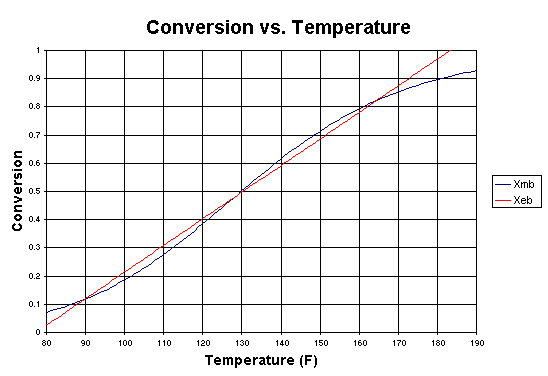
Figure CDE13-3.1 |
|
|
||
Based on a steady-state analysis, it would appear that after
the drop in the inlet temperature we should remain at the upper steady state
with T = 164.5°F and X = 82.3%. However, using a dynamic
simulation (see the development in the equations below) with POLYMATH (Table CDE13-3.3), we will see that with this perturbation in the inlet temperature, the conversion and temperature drop to the lower steady-state values, as shown in Figure CDE13-3.2.
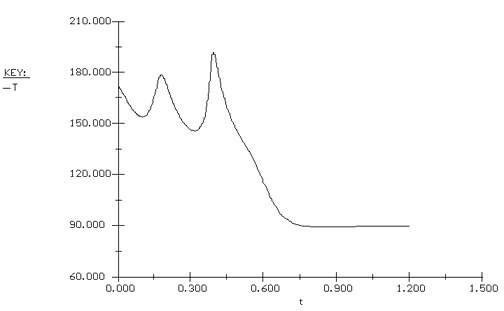
Temperature Time Trajectory |
||
Unsteady-State Equations After Temperature Perturbation |
||
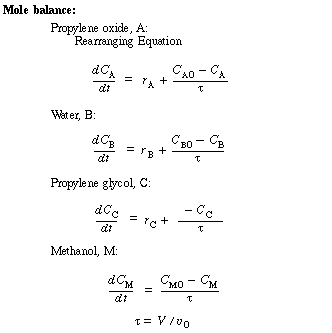
|
||
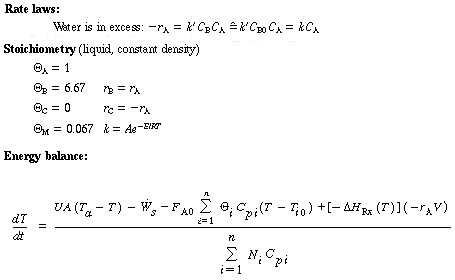
|
||
For constant volume, constant heat of reaction, and no work done by the system, when applied to this example becomes |
||
|
|
||
The number of moles Ni in the denominator is just Ni = CiV. |
||
|
|
|
|
We see that even though the entering temperature did not drop below the extinction temperature, the perturbation in the entering temperature was large enough to drive the temperature and conversion drop to their lower steady-state values of 89.5°F and 11.6%, respectively. To prevent this problem of moving to an undesired steady state, a control system is usually incorporated into the reactor system. |