Chapter 14: External Diffusion Effects on Heteregeneous Reactions
Additional Homework Problems
CDP14-KB
In a certain chemical plant, a reversible fluid-phase isomerization
A 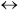 B
B
is carried out over a solid catalyst in a tubular packed-bed reactor. If the reaction is so rapid that mass transfer between the catalyst surface and the bulk fluid is rate-limiting, show that the kinetics are described in terms of the bulk concentrations CA and CB by
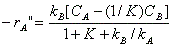
where -r"A = moles of A reacting per unit area catalyst per time
kA, kB = mass transfer coefficients for A and B
K = reaction equilibrium constant
It is desired to double the capacity of the existing plant by processing twice the feed of reactant A while maintaining the same fractional conversion of A to B in the reactor. How much larger a reactor, in terms of catalyst weight, would be required if all other operating variables are held constant? You may use the Thoenes-Kramaers correlation for mass transfer coefficients in a packed bed. Describe the effects of the flow rate, temperature, and particle size on conversion.
[3rd Ed. P11-6]