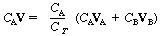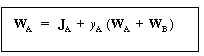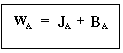Chapter 14: External Diffusion Effects on Heteregeneous Reactions
Learning Resources
Example CD14-2: Relating the Fluxes WA, BA, and JA
| Derive Equation (11-6) starting from the fact that the molecular diffusional flux is the flux relative to the molar average velocity. | |||
Solution |
|||
| The difference in velocities (VA -V) tells us the velocity at which species A is moving relative to the average velocity of all the species. The diffusive flux JA of A relative to the molar average velocity is | |||
|
Velocity of A |
JA = CA
(VA -V) |
(CDE11-2.1) (CDE11-2.2) |
|
| Using Equation (11-3) yield | |||
|
JA = WA -CAV |
(CDE11-2.3) | ||
| Rearranging gives us | |||
|
WA = JA +CAV |
(CDE11-2.4) | ||
| The second term on the right-hand side can be expanded to | |||
|
CAV = CA(yAVA + yBVB) |
(CDE11-2.5) | ||
| Factoring the reciprocal of the total concentration, 1/CT, from the term in parentheses, we have | |||
|
|
(CDE11-2.6) | ||
| Then | |||
|
|
(11-6) | ||
Recalling that BA = CAV = YA (WA + WB
|