|
|
The Enemark-Feltham Notation The Enemark-Feltham notation is frequently used to keep track of the oxidation states of transition-metal nitric oxide (NO) complexes [1]. This is due to the fact
that NO is a non-innocent ligand that can become oxidized (to NO+) or reduced (to NO-) when binding to a transition metal center, M. In addition, the bond between NO and transition-metals
is often times very covalent, making it impossible to know what the electron distribution in the M-N-O unit is without performing detailed spectroscopic and theoretical studies.
In order to deal with this complication, it is therefore convenient to simply consider the MNO unit as a covalent entity, and to just count the number of valence electrons of the MNO unit.
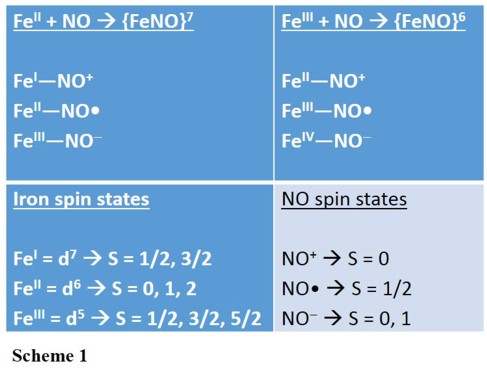
In the Enemark-Feltham notation, {M(NO)x}n, the index x indicates the number of coordinated NO molecules to metal M, and the exponent n counts the number of valence
electrons, which is the number of metal(d) and NO(π*) electrons. For example, the adduct of an Fe(II) complex and NO would therefore be classified as {FeNO}7, where the exponent '7' is the
sum of six Fe(d) and one NO(π*) electrons. Scheme 1 shows the different electron distributions that are possible for {FeNO}7 and {FeNO}6 complexes. Literature: |
| OPEN POSITIONS |
HOME |
RESEARCH |
METHODS |
PUBLICATIONS |
CV |
LECTURES |
GROUP MEMBERS |