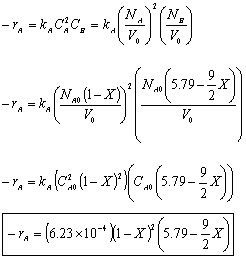Chapter 4: Stoichiometry
Oxidation of Naphthalene to Phtahalic Anhydride
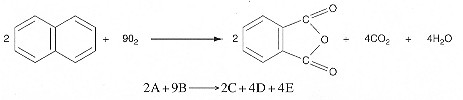
We are going to rework the class problem for the case of 3.5% naphthalene and 96.7% air and for a pressure of 10 atm and a temperature of 500K. A feed under these conditions naphthalene is the limiting reactant.
- Stoichiomentric Table - Batch System
Stoichiometric Table - Flow SystemSpecies Symbol In Change Out Naphthalene A NA0 -NA0X NA = NA0(1-X) Oxygen B NB0 = 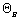 NA0
NA0-9/2NA0X NB = NA0( 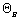 - 9/2X)
- 9/2X)Phthalic Anhydride C NC = 0 +NA0X NC = NA0X Carbon Dioxide D ND = 0 +2NA0X ND = 2NA0X Water E NE = 0 +2NA0X NE = 2NA0X Nitrogen I NI = 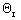 NA0
NA0--- NI = 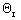 NA0
NA0Total NT0 NT = NT0 + 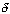 NA0X
NA0X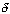 = [1 + 2 + 2 - 9/2 - 1] = -1/2
= [1 + 2 + 2 - 9/2 - 1] = -1/2Species Symbol In Change Out Naphthalene A FA0 -FA0X FA = FA0(1-X) Oxygen B FB0 = 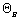 FA0
FA0-9/2FA0X FB = FA0( 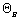 - 9/2X)
- 9/2X)Phthalic Anhydride C FC = 0 +FA0X FC = FA0X Carbon Dioxide D FD = 0 +2FA0X FD = 2FA0X Water E FE = 0 +2FA0X FE = 2FA0X Nitrogen I FI = 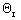 FA0
FA0--- FI = 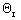 FA0
FA0Total FT0 FT = FT0 + 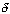 FA0X
FA0X 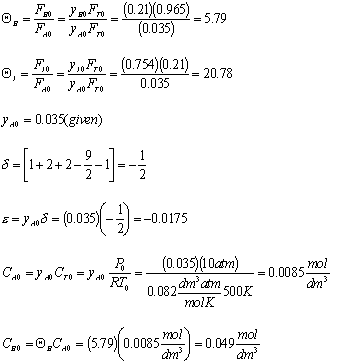
- Determine each of the following solely as a function of the conversion of naphthalene, X for a constant-pressureisothermal flow reactor.
- Find the concentration of O2
Gas Phase Flow System:
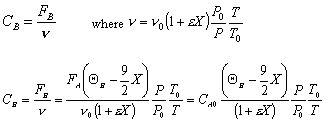
Constant pressure and isothermal: P = Po; T = To
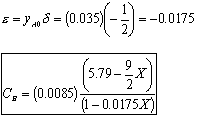
- Find the volumentric flowrate
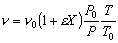
Constant pressure and isothermal: P = Po; T = To
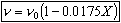
- Find the reaction rate
Rate Law: -rA = kAC2ACB
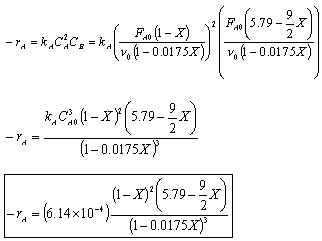
- Find the concentration of O2
- The concentration of O2
For Batch: V = V0, CB = NB/V = NB/V0
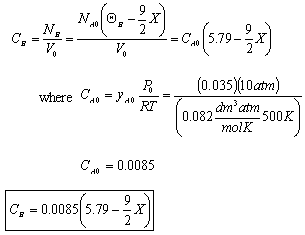
- The total pressure, P.
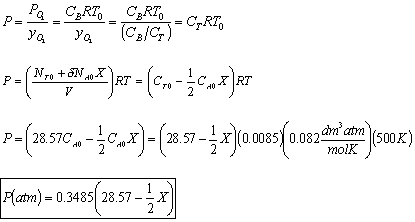
- The rate law
Rate Law: -rA = kAC2ACB