Chapter 6: Isothermal Reactor Design: Molar Flow Rates
Additional Homework Problems
CDP6-FA
The removal of nitrogen from organic compounds (i.e., hydrodenitrogenation)
is an important industrial reaction. We consider the first reaction step of this
liquid-phase process in which 5,6-benzoquinoline (species A) is reacted at 100°C
in a solution saturated with hydrogen (2500 psig) (i.e., excess H 2
) [Ind. Eng. Chem., 28, 139 (1989)].
The following data were obtained at 100°C in a batch reactor using sulfided
CoMo as a catalyst at a concentration of 20 g/dm 3
.
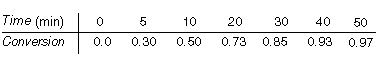
Also note that the rate at 110° C is approximately
four times the rate at 80°C. Verify that the reaction is pseudo-first order
in 5,6-benzoquinoline and determine the specific reaction rate.
It has been learned that the specific reaction rate is directly proportional to the
catalyst concentration (i.e., first order). It is proposed to double the catalyst
concentration and drop the temperature to 90°C. Plot the conversion expected
under these conditions as a function of time and compare with the data above.
[2nd Ed. P4-7]