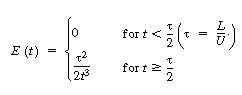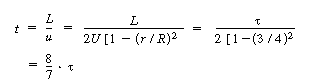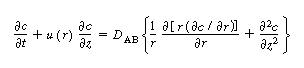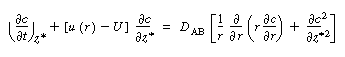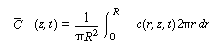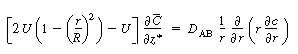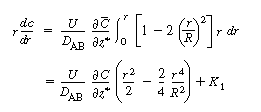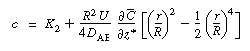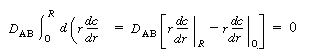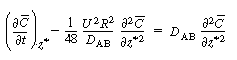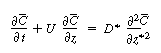Chapter 18: Models for Nonideal Reactors
Professional Reference Shelf
R18.1 Derivation of Equation for Taylor-Aris Dispersion
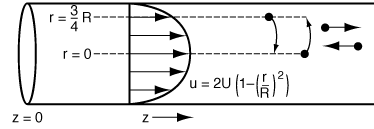
| In a laminar flow reactor we know that the axial velocity varies in the radial direction according to the Hagen-Poiseuille equation: | |||
|
|
|||
| where U is the average velocity. For laminar flow we saw that the RTD function E(t) was given by | |||
|
|
|||
| In arriving at this distribution E(t) it was assumed
that there was no transfer of molecules in the radial direction between streamlines.
Consequently, with the aid of Equation (13-43), we know that the molecules on the
center streamline (r = 0) exited the reactor at a time t = |
|||
|
|
(13-43) | ||
| The question now arises: What would happen if some of the molecules traveling on the streamline at r = 3R/4 jumped (i.e., diffused) to the streamline at r = 0? The answer is that they would exit sooner than if they had stayed on the streamline at r = 3R/4. Analogously, if some of the molecules from the faster streamline at r = 0 jumped (i.e., diffused) to the streamline at r = 3R/4, they would take a longer time to exit (Figure R14.1-1). In addition to the molecules diffusing between streamlines, they can also move forward or backward relative to the average fluid velocity by molecular diffusion (Fick's law). With both axial and radial diffusion occurring, the question arises as to what will be the distribution of residence times when molecules are transported between and along streamlines by diffusion. To answer this question we will derive an equation for the axial dispersion coefficient, D a , that accounts for the axial and radial diffusion mechanisms. In deriving D a , which is referred to as the Aris-Taylor dispersion coefficient, we closely follow the development given by Brenner and Edwards. 1 | |||
|
|
|||
| The convective-diffusion equation for solute (e.g., tracer) transport in both the axial and radial direction is | |||
|
|
(R14.1-1) | ||
| We are going to change the variable in the axial direction z
to |
|||
|
|
(R14.1-2) | ||
| A value of |
|||
|
|
(R14.1-3) | ||
| Because we want to know the concentrations and conversions at the
exit to the reactor, we are really only interested in the average axial concentration
|
|||
|
|
(R14.1-4) | ||
| Consequently, we are going to solve Equation (R14.1-3) for the solution
concentration as a function of r and then substitute the solution c(r,
z, t) into Equation (R14.1-4) to find To solve equations (R14.1-1) to (R14.1-4) to determine the Aris-Taylor dispersion coefficient, we make the following four assumptions: |
|||

|
|||
| We now apply the preceding approximations to Equation (R14.1-4) to arrive at the following equation: | |||
|
|
(R14.1-5) | ||
| Because |
|||
|
|
|||
| Symmetry conditions dictate that at r = 0, After dividing both sides by r, a second integration yields |
|||
|
|
(R14.1-6) | ||
| The constant of integration K 2 is independent of r and can be evaluated using the equation for the average concentration: | |||
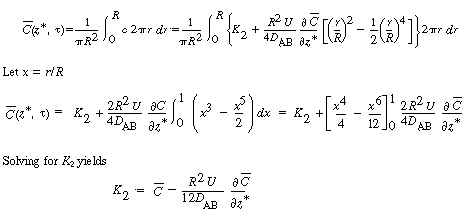 |
|||
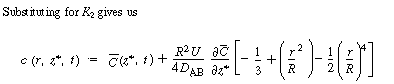 |
(R14.1-7) | ||
| The final step to arrive at the celebrated Aris-Taylor dispersion
coefficient,D*, is to multiply Equation (R14.1-3) by
the differential cross-sectional area 2 |
|||
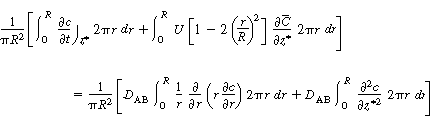
|
|||
| By changing the order of differentiation and integration for the first and last terms and recalling Equation (R14.1-4) gives | |||
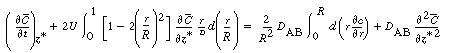
|
(R14.1-8) | ||
| Because there is no transport of solute through the tube wall, | |||
|
|
|||
| Integrating the first term on the right-hand side of Equation (R14.1-8) along with the preceding boundary condition above gives | |||
|
|
(R14.1-9) | ||
| Next we differentiate Equation (R14.1-7) with respect to |
|||
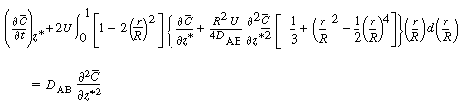 |
(R14.1-10) | ||
| Carrying out the integration between 0 and 1 (r = R) gives | |||
|
|
(R14.1-10) | ||
| Changing our variable |
|||
|
|
(R14.1-11) | ||
| where D* is the Aris-Taylor dispersion coefficient: | |||
|
Aris-Taylor dispersion coefficient |
|
(R14.1-12) | |
| That is, for laminar flow in a pipe | |||
|
|
|||
| Figure 14.5 shows the dispersion coefficient D* in terms of
the ratio |
|||