Compute-to-Learn
The Compute-to-Learn Pedagogy: Developing interactive, computer-based demonstrations

The Compute-to-Learn pedagogy engages students in authentic scientific practices
as they construct interactive, computer-based demonstrations of physical chemistry
concepts. This pedagogy was implemented during the Fall 2015 semester within the
Chem 260 Honors Studio, a peer-led studio supplement to an introductory physical
chemistry course. Students in the honors studio design and develop interactive
demonstrations using the Mathematica software package, and ultimately submit these
demos for publication as part of the Wolfram Demonstrations Project.
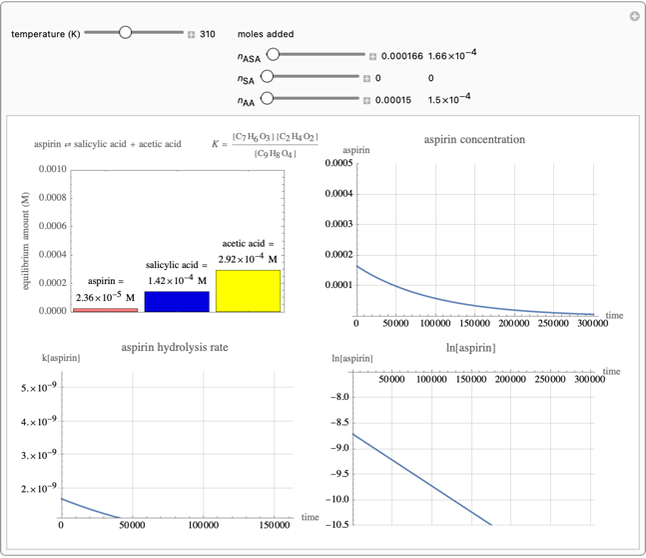
The Geva group has developed a number of interactive software demonstrations in
conjuction with the Wolfram Demonstrations Project, which Professor Geva has
incorporated into many of his lectures. These software applications provide students
with a dynamic graphical representation of various physical chemistry concepts,
allowing them to adjust various parameters and instantly observe the impact. An
example of one of these software demonstrations is displayed below, and the complete
list of Professor Geva's Mathematic demos can be viewed here.
The Wolfram Demonstrations Project is an open-code resource that uses dynamic computation
to illuminate concepts in science, technology, mathematics, art, finance, and a
remarkable range of other fields. All Demonstrations run freely on any standard Windows,
Mac, or Linux computer.
Courses Professor Geva has taught
- Chemistry 260 - Chemical Principles / Chemistry 261 - Introduction to Quantum Chemistry
Introduction to the quantal nature of matter, basic chemical thermodynamics, and chemical kinetics. Description of the fundamental physical principles that underlie spectroscopic and electrochemical analysis. CHEM 261 is an introduction to the quantal nature of matter (the Schrödinger equation and the mathematical machinery of quantum mechanics) and the fundamental principles necessary to understand spectroscopy (electronic, vibrational, and rotational). This course, together with CHEM 330, provides the prerequisites necessary for enrollment in CHEM 302.
- Chemistry 453 - Biophysical Chemistry I: Thermodynamics and Kinetics
First in a two-term biophysical chemistry sequence for biochemistry students. Emphasis on topics and applications relevant to biochemistry and modern biophysical chemistry, building on CHEM 260. Rigorous mathematical theory of classical thermodynamics is developed, including application to entropy, heat engines, solution properties, and phase and chemical equilibrium. Modern statistical thermodynamics, modern theories of fundamental reaction rates and enzyme kinetics and molecular transport theories are described and developed.
- Chemistry 461 - Physical Chemistry I
This is the second of the three-term physical chemistry sequence CHEM 260/461/463. CHEM 461 builds on the introduction to quantum mechanics that was given in CHEM 260. Students will use the Schrödinger Equation in 1-, 2-, and 3 dimensions to solve exactly a series of important chemical problems including the harmonic oscillator, the rigid rotor, and the hydrogen atom. Group theory is introduced as an aid for understanding spectroscopic selection rules. Advanced spectroscopy, including transition probabilities, normal vibrational modes, and photoelectron spectroscopies are introduced and then used to deduce molecular structure. The valence-bond and molecular orbital theories of chemical bonding are discussed, and methods for performing quantum chemical calculations, including variational and perturbation methods, are introduced. The quantum mechanics of spin and angular momentum are discussed and used to interpret magnetic resonance spectra.
- Chemistry 567 - Chemical Dynamics
Chemical Kinetics is the study of the rates and mechanisms of systems undergoing chemical change. The extraction of rate data from reacting systems and the utilization of such data in other reacting systems is central to chemistry in the laboratory and in the practical worlds of combustion science, atmospheric science, and chemical synthesis. This course introduces the treatment of complex chemical systems and fundamental ideas about chemical reaction rates in gases and in solutions. Computer software is utilized to treat complex reaction systems.
- Chemistry 571 - Quantum Chemistry
Constitutes with CHEM 576 a full course for students specializing in physical chemistry. Review of quantum mechanics from a postulational viewpoint; variational and matrix methods, time-independent and time-dependent perturbation theory; applications to molecular systems including potential energy surfaces and reaction pathways.
- Chemistry 580 - Molecular Spectra and Structure
CHEM 580 is an advanced physical chemistry graduate course on molecular structure, dynamics and spectroscopy.
An Electronic Structure Tutorial for Undergraduate Research Students
by Heidi Phillips Hendrickson
Phillips, H. (2014, August). Designing an authentic and interactive tutorial on quantum chemistry for undergraduate researchers: An apprenticeship model. Paper presented at the Biennial Conference on Chemical Education, Allendale, MI.
The following abstract provides a brief description of the approach I used to design an electronic structure tutorial for undergraduate students in the Geva and Dunietz research groups. Please feel free to download the presentation slides for your reference.
Designing an Authentic and Interactive Tutorial on Quantum Chemistry for Undergraduate Researchers: An Apprenticeship Model
This presentation will describe an interactive tutorial aimed at teaching undergraduate research students how to use computational chemistry software through authentic research activities. Based on an apprenticeship model (Lave & Wenger, 1991), the tutorial combines multiple principles for designing successful science learning environments including an emphasis on key concepts and collaboration (Kracjik, Slotta, McNeill, & Reiser, 2008). The interactive tutorial consists of instructional lectures, interactive and follow-up exercises with associated input files, and documentation to guide the instructor. The exercises emphasize a combination of quantum chemistry concepts and technical skills, requiring students to not only create and modify files in challenging ways, but to analyze the results of their calculations within the context of quantum chemistry. While the main focus is for undergraduate students planning to engage in computational chemistry research, the tutorial package could also be modified for inclusion in a physical chemistry course. The tutorial and related artifacts were designed within the STEM (Science, Technology, Engineering, and Math) Studio at the University of Michigan, which provides a trans-disciplinary workshop environment for improving STEM-related educational materials.
References
- Lave, J., & Wenger, E. (1991). Situated learning: Legitimate peripheral participation. New York, NY US: Cambridge University Press.
- Krajcik, J. S., Slotta, J. D., McNeill, K. L., & Reiser, B. J. (2008). Designing learning environments to support students’ integrated understanding. In Y. Kali, M. Linn, & J. Roseman (Eds.), Designing coherent science education (pp. 39–64). New York, NY: Teachers College Press.